40. Immune Receptor Profiling#
Key takeaways
VDJ-sequencing measures the sequence information of AIRs, which provides us insights into the function of B- and T-cells.
To detect doublets, cells with multiple AIRs should be identified or filtered.
Depending on the analysis, we additional filter cells with incomplete AIR information.
Environment setup
Install conda:
Before creating the environment, ensure that conda is installed on your system.
Save the yml content:
Copy the content from the yml tab into a file named
environment.yml.
Create the environment:
Open a terminal or command prompt.
Run the following command:
conda env create -f environment.yml
Activate the environment:
After the environment is created, activate it using:
conda activate <environment_name>
Replace
<environment_name>with the name specified in theenvironment.ymlfile. In the yml file it will look like this:name: <environment_name>
Verify the installation:
Check that the environment was created successfully by running:
conda env list
40.1. Role IR in cells#
Immune Receptors (IR) are the key for the recognition of potential hazardous antigens and toxins invading the body. Those receptors are usually located in the membrane of specialized cells, which recognize harmful content and initiate protective mechanisms. There are different kinds of IRs specialized in the recognition of specific structures of foreign agents:
Pattern recognition receptors (PRRs): Pathogen-associated molecular patterns (PAMPS)
Killer activator/inhibitor receptors (KARs/KIRs): Host cells abnormalities
Complement receptors: Complement proteins
Fc receptors: Epitope-antibody complex
Cytokine receptors: Cytokines
B-cell receptor (BCRs): Epitopes
T-cell receptors (TCRs): Linear epitopes bound to the Major Histocompatibility Complex (MHC)
40.2. IRs of the adaptive immune system#
In the adaptive immune system, there are two major lineages of lymphocytes with IRs, which are produced in the Thymus and Bone-marrow called T- and B-cells [Cooper and Alder, 2006]. These adaptive immune receptors (AIRs) of both cell lines detect antigens derived from different pathogens and tumor cells. However, they interact with the antigens in different ways: While the B-cell receptor (BCR) directly recognizes soluble or membrane-bound epitopes, the T-cell receptor interacts with linear peptides bound to the surface protein MHC (pMHC), which enables to view inside cells by presenting its content on the cell’s surface. After activation by antigens, B- and T-cells perform various functions such as fighting the pathogen, regulation of the immune response, or forming memory by proliferation.
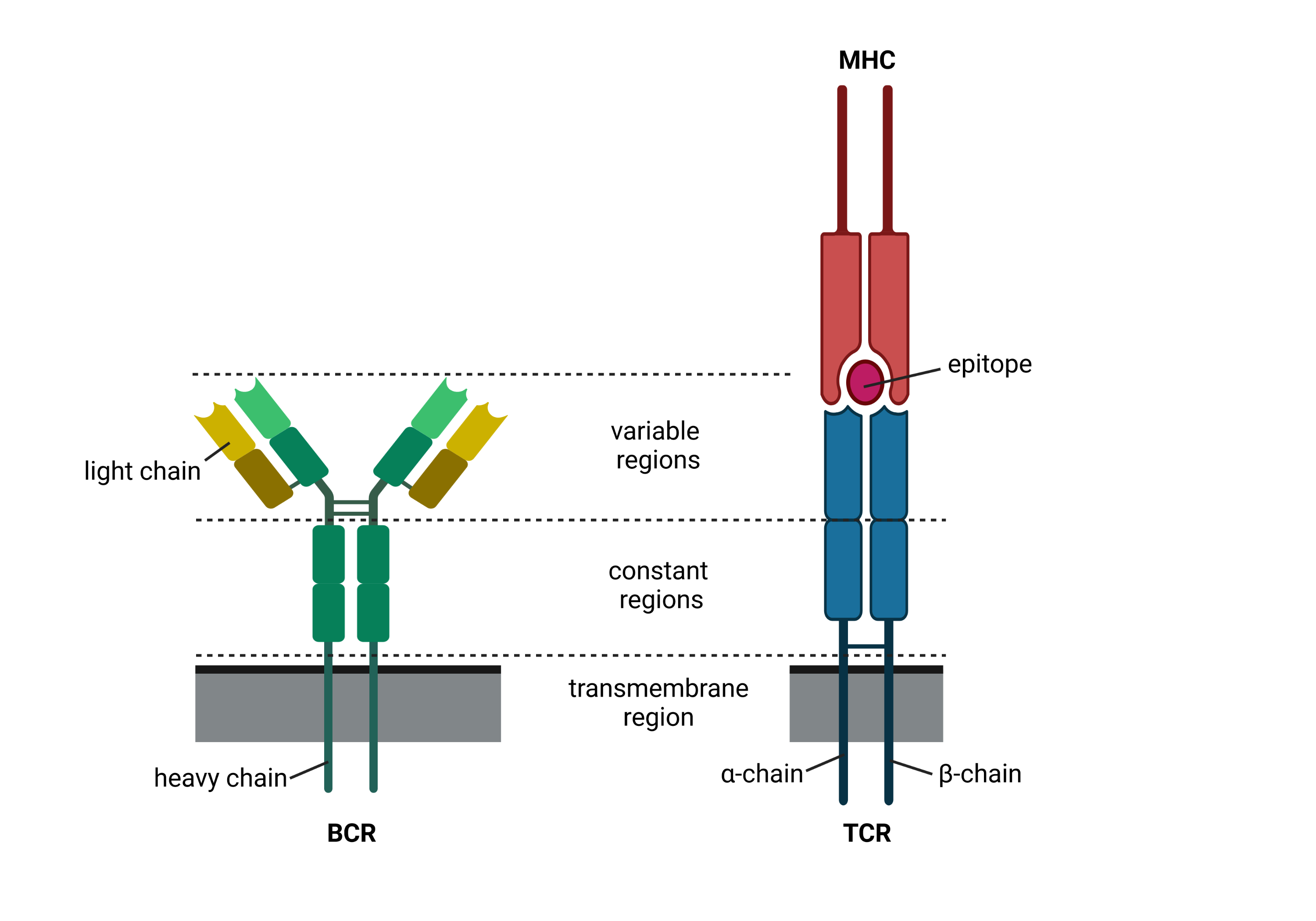
Fig. 40.1 Schematic drawing of the two AIR types. Created with BioRender.com.#
40.3. Formation of adaptive immune receptors by V(D)J recombination#
The AIR is a protein complex consisting of two chains. Depending on the T cell type, these chains are either called α- and β- or γ- and δ-chain. Similarly, the BCR chains are called heavy and light chain (subtypes: κ and λ). While the α-, γ-, κ-, and λ-chains are formed by their respective V- and J-genes, the remaining chains additionally include their D-gene. For simplicity, we will therefore refer to VJ and VDJ chains throughout this book.
Adaptive IRs lines are highly polymorphic in order to capture the immense space of antigens, i.e., there exists an immense variety of different TCR and BCR sequences. The amount of different TCRs have been estimated at 1020 in total, while a human individual harbors 107 different sequences [Zarnitsyna et al., 2013]. Similarly, there naturally occur up to 1018 different BCRs [Briney et al., 2019]. The collection of all receptors forms the AIR repertoire of an individual.
The sequence diversity is introduced by the V(D)J recombination mechanism:
Combinatorial diversity is introduced by the combination of different V-, (D-,) and J-genes in addition to the combination of different VJ and VDJ chains.
Junctional diversity is achieved by insertion of nucleotides at the gene interfaces.
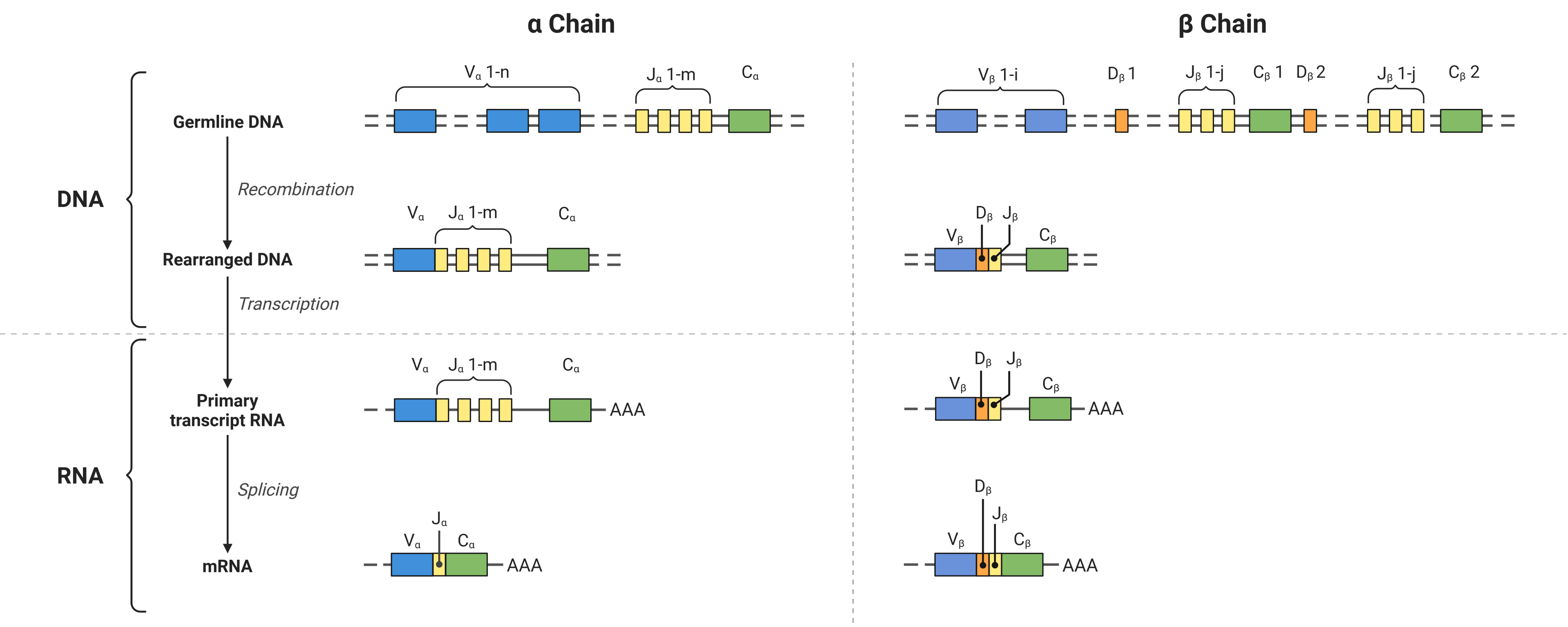
Fig. 40.2 VDJ recombination at the example of an αβ-TCR. Created with BioRender.com.#
Additionally, mutations occur at the BCR binding sites in a process called Somatic Hypermutation for affinity maturation during rapid cell proliferation.
In the resulting AIRs, three regions of high variability - the Complementary Determining Regions (CDRs) 1-3 - were detected per chain, where the AIR interacts with its target. The CDR1 and CDR2 are encoded by the V-gene. However, the CDR3 spans over the intersection of V-, (D-,) and J-genes and is therefore the most diverse element of the AIR. Therefore, it is often assumed that the specificity of an AIR is determined mainly by its CDR3 region of the more diverse VDJ chain.
40.4. VDJ-sequencing#
40.4.1. Cell isolation#
Before quantifying the single-cell data, the cells of interest should be targeted and isolated. Common options include:
Fluorescence Activated Cell Sorting (FACS) is a method based on flow cytometry with the power to label the cells of interest based on fluorescent probes over the raw cell suspension. A cell suspension is carried by a rapidly flowing stream of liquid. This stream of cells is broken up into individual droplets through a vibrating mechanism. Just before the stream breaks into droplets, the flow passes through a fluorescence measuring station where the fluorescence signal of every cell is measured. The droplets can be further charged for further separations.
Magnetic-Activated Cell Sorting (MACS) can use antibodies, enzymes, lectins, or streptavidins attached to a magnetic bead to label the target cells. Once the cells are labeled from the raw suspension, a magnetic field is applied to attract the magnetic beads and discard the remaining cells from the suspensions. The targeted cells are collected once the magnetic field is turned off. One advantage of this method is the capacity to collect targeted cells with no specific markers to be labeled, in that case, a cocktail of markers is used to label the untargeted cells, and the cells of interests are collected by washing them out once the magnetic field captures the untargeted cells conjugated to a magnetic bead.
Laser Capture Microdissection (LCM) has the power to extract cell populations or single cells from microscope preparations without detriment of the surrounding tissue. The components to perform LCM includes a reverse microscope, a laser control unit, a microscope joy stick to plate stabilization, a CCD camera, and a color monitor. The idea behind LCM consists on labelling cells by visual detection of morphological characteristics of target cells, the plate is immobilized and the laser pulse melts the thin thermoplastic film removing the cells or cells of interest without any damage to the surrounding tissue.
Microfluidics is a versatile method able to work with small quantities of raw suspension even at the order of nanoliters. There are different kinds of microfluidic approaches including cell-affinity chromatography based microfluidics, physical characteristics of cell based microfluidics, immunomagnetics beads based microfluidics, and separation by dielectric properties of some cell-types based microfluidics. The most used microfluidics based method is the chromatographic separation using a chip assay as stationary phase which is modified to include the necessary antibodies to capture the target cells in the mobile phase. After the buffer flows off from the chip, a solution is used to separate the cells attached to the antibodies to collect them for further analysis [Hu et al., 2016].
40.4.2. Immune receptor sequencing#
A common approach to discern V(D)J chains from single-cell isolations consists of computational reconstructions of different chains’ sequences based on full-length single-cell RNA sequencing, with Smart-seq2, a 5’-end RNA template based protocol, being one of the most widely implemented. Regarding computational methods, TRAPeS, TraCer, and VDJPuzzle are usually used to reconstruct TCR sequences based on scRNA-seq data, whereas BALDR [Upadhyay et al., 2018], BASIC [Canzar et al., 2016] and BraCer [Lindeman et al., 2018] were shown to robustly recover BCR sequences. However, they are prone to ignore the whole landscape of recombinatorial products and alternative splicing products in V(D)J region. Some alternatives have arisen to deal with this problem, RAGE-seq for example was developed to capture specific TCR and BCR fragments based on PCR templates designed for immune receptor sequencing and use long-read Oxford Nanopore to capture the whole sequence, whereas the rest of the cDNA is processed based on short-reads protocols provided by, for example, Illumina [Singh et al., 2019].
40.5. AIR repertoire analysis#
VDJ-sequencing provides us with the nucleotide and thereby also the protein sequence of the AIR paired for both chains, from which the V-, (D-,) J-, and C-gene is determined in addition to the CDR3 sequence. Overall, the AIR sequence determines the specificity of the individual B- and T-cell. Therefore, the information obtained by VDJ-sequencing provides us with an indicator of the cells’ functionality, which is directly coupled to the AIRs target antigen. This enables us to use the AIR information in three major ways:
Phenotyping: We can group immune cells by identifying cells with the same or similar AIR, which share the same specificity. Having these groups, we can now observe, how disease-specific cells react under different conditions (e.g. transcriptomic change upon stimulation), whether immune cells have proliferated, or how the diversity of an immune repertoire changes after an immune response.
Sequence Analysis: Having identified groups of AIRs (e.g. a reactive cluster detected in other modalities), we can extract properties of their sequence, such as V-, D-, and J-, gene usage or enriched sequence motifs, that are related to specific diseases or therapies.
Specificity-Inference: Last, we can use the sequence to match AIRs to their target antigen via database queries, sequence distances, or predictors. This directly identifies cells reactive to specific infectious diseases, tumors, or self-antigens.
40.6. Dataset#
To showcase different approaches for preprocessing and analysis of IRs, we will use the dataset published by the Haniffa Lab [Stephenson et al., 2021], which contains transcriptome sequencing for over 750,000 Peripheral Blood Mononuclear Cells (PBMC) from 130 patients in the context of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2).
It consists of patients from three sources (Newcastle, Cambridge, and London) with different degrees of severity (asymptomatic, mild, moderate, severe, and critical) as well as negative controls (healthy, other severe respiratory illnesses, and healthy with Intravenous Liposaccharide to mimic systemic inflammatory response). Additional, patient-level information such as age, gender, and smoker status are provided.
The dataset was chosen as an example of a large-scale single cell dataset including VDJ-Sequencing, that is well known to the community. It contains over 150,000 B-Cells and 200,000 T-Cells with IR annotation, that we will regard in this analysis tutorial.
To download the data please run the following commands:
path_data = "data/"
path_bcr_input = f"{path_data}/BCR_00_read_aligned.csv"
path_tcr_input = f"{path_data}/TCR_00_read_aligned.tsv"
! wget -O $path_bcr_input -nc https://figshare.com/ndownloader/files/35574338
! wget -O $path_tcr_input -nc https://figshare.com/ndownloader/files/35574539
--2022-06-07 18:30:52-- https://figshare.com/ndownloader/files/35574338
Resolving figshare.com (figshare.com)... 54.72.163.193, 52.50.42.102, 2a05:d018:1f4:d000:7421:2135:71b2:6a10, ...
Connecting to figshare.com (figshare.com)|54.72.163.193|:443... connected.
HTTP request sent, awaiting response... 302 Found
Location: https://s3-eu-west-1.amazonaws.com/pfigshare-u-files/35574338/bcr_cellranger.csv?X-Amz-Algorithm=AWS4-HMAC-SHA256&X-Amz-Credential=AKIAIYCQYOYV5JSSROOA/20220607/eu-west-1/s3/aws4_request&X-Amz-Date=20220607T163052Z&X-Amz-Expires=10&X-Amz-SignedHeaders=host&X-Amz-Signature=b18cb63ca03820e07cb8b3d9375466ca6405e655822269f5ae36d7449a28b810 [following]
--2022-06-07 18:30:52-- https://s3-eu-west-1.amazonaws.com/pfigshare-u-files/35574338/bcr_cellranger.csv?X-Amz-Algorithm=AWS4-HMAC-SHA256&X-Amz-Credential=AKIAIYCQYOYV5JSSROOA/20220607/eu-west-1/s3/aws4_request&X-Amz-Date=20220607T163052Z&X-Amz-Expires=10&X-Amz-SignedHeaders=host&X-Amz-Signature=b18cb63ca03820e07cb8b3d9375466ca6405e655822269f5ae36d7449a28b810
Resolving s3-eu-west-1.amazonaws.com (s3-eu-west-1.amazonaws.com)... 52.218.62.171
Connecting to s3-eu-west-1.amazonaws.com (s3-eu-west-1.amazonaws.com)|52.218.62.171|:443... connected.
HTTP request sent, awaiting response... 200 OK
Length: 84451806 (81M) [text/csv]
Saving to: ‘data//BCR_00_read_aligned.csv’
100%[======================================>] 84,451,806 39.1MB/s in 2.1s
2022-06-07 18:30:54 (39.1 MB/s) - ‘data//BCR_00_read_aligned.csv’ saved [84451806/84451806]
--2022-06-07 18:30:55-- https://figshare.com/ndownloader/files/35574539
Resolving figshare.com (figshare.com)... 54.72.163.193, 52.50.42.102, 2a05:d018:1f4:d000:7421:2135:71b2:6a10, ...
Connecting to figshare.com (figshare.com)|54.72.163.193|:443... connected.
HTTP request sent, awaiting response... 302 Found
Location: https://s3-eu-west-1.amazonaws.com/pfigshare-u-files/35574539/TCR_mergedUpdated.tsv?X-Amz-Algorithm=AWS4-HMAC-SHA256&X-Amz-Credential=AKIAIYCQYOYV5JSSROOA/20220607/eu-west-1/s3/aws4_request&X-Amz-Date=20220607T163055Z&X-Amz-Expires=10&X-Amz-SignedHeaders=host&X-Amz-Signature=141ab989ae794394fb6c441d50c0ea6771f96a8d048a8a4c400c10dba267b97c [following]
--2022-06-07 18:30:55-- https://s3-eu-west-1.amazonaws.com/pfigshare-u-files/35574539/TCR_mergedUpdated.tsv?X-Amz-Algorithm=AWS4-HMAC-SHA256&X-Amz-Credential=AKIAIYCQYOYV5JSSROOA/20220607/eu-west-1/s3/aws4_request&X-Amz-Date=20220607T163055Z&X-Amz-Expires=10&X-Amz-SignedHeaders=host&X-Amz-Signature=141ab989ae794394fb6c441d50c0ea6771f96a8d048a8a4c400c10dba267b97c
Resolving s3-eu-west-1.amazonaws.com (s3-eu-west-1.amazonaws.com)... 52.218.62.171
Connecting to s3-eu-west-1.amazonaws.com (s3-eu-west-1.amazonaws.com)|52.218.62.171|:443... connected.
HTTP request sent, awaiting response... 200 OK
Length: 247085830 (236M) [text/tab-separated-values]
Saving to: ‘data//TCR_00_read_aligned.tsv’
100%[======================================>] 247,085,830 42.4MB/s in 5.8s
2022-06-07 18:31:01 (40.9 MB/s) - ‘data//TCR_00_read_aligned.tsv’ saved [247085830/247085830]
40.7. Load data#
In this tutorial we will mainly use two python packages for loading, cell-level ordering, and visualization:
Scanpy: general package for single cell analysis (theislab/scanpy, [Wolf et al., 2018])
Scirpy: scanpy extension for immune receptor analysis (scverse/scirpy, [Sturm et al., 2020])
Here, we only showcase IR analysis with Scirpy. However, there exist several tools with similar functionality such as immunarch(R, [ImmunoMind Team, 2019]), scRepertoire (R, [Borcherding et al., 2020]), and dandelion (R, [Stephenson et al., 2021]), and Platypus (R, [Yermanos et al., 2021]) reviewed in [Valkiers et al., 2022].
Warning
Scirpy changed the format of its datastructure with v0.13. While the overall analysis workflow has not changed, some outputs shown in this chapter might not be accurate anymore.
See the scirpy release notes for more details about this change. Until we update this chapter, please also refer to the official scirpy documentation.
import warnings
warnings.filterwarnings(
"ignore",
".*IProgress not found*",
)
warnings.simplefilter(action="ignore", category=FutureWarning)
import pandas as pd
import scanpy as sc
import scirpy as ir
warnings.simplefilter(action="ignore", category=pd.errors.DtypeWarning)
Let’s set the input and output paths of our data.
path_bcr_out = f"{path_data}/BCR_01_preprocessed.h5ad"
path_tcr_csv = f"{path_data}/TCR_00_read_aligned.csv"
path_tcr_out = f"{path_data}/TCR_01_preprocessed.h5ad"
40.7.1. Raw data#
We begin by viewing the raw output of the cell ranger pipeline for a better understanding of the data we are working with.
We will load the filtered_contig_annotations.csv" file to view its content. Each row will represent one measurement of a sequence.
df_bcr_raw = pd.read_csv(path_bcr_input, index_col=0)
# The column 'productive' contains mixed data types which are not compatible with downstream tools.
# We correct them by casting them to strings.
df_bcr_raw["productive"] = df_bcr_raw["productive"].astype(str)
print(f"Total measurements: {len(df_bcr_raw)}")
df_bcr_raw.head(5)
Total measurements: 373670
| barcode | is_cell | contig_id | high_confidence | length | chain | v_gene | d_gene | j_gene | c_gene | full_length | productive | cdr3 | cdr3_nt | reads | umis | raw_clonotype_id | raw_consensus_id | patient_id | newBarcode | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 | AAACCTGAGCGATATA-MH9143270 | True | AAACCTGAGCGATATA-MH9143270_contig_1 | True | 646 | IGL | IGLV1-44 | None | IGLJ1 | IGLC2 | True | True | CAAWDDSLSASYVF | TGTGCAGCATGGGATGACAGCCTGAGTGCCTCTTATGTCTTC | 154 | 14 | clonotype9 | clonotype9_consensus_2 | COVID-002 | NaN |
| 1 | AAACCTGAGCGATATA-MH9143270 | True | AAACCTGAGCGATATA-MH9143270_contig_2 | True | 579 | IGH | IGHV3-33 | IGHD3-3 | IGHJ4 | IGHM | True | True | CARDNRVYYDFWSGYPDYW | TGTGCGAGAGACAACCGAGTCTATTACGATTTTTGGAGTGGTTATC... | 144 | 13 | clonotype9 | clonotype9_consensus_1 | COVID-002 | NaN |
| 2 | AAACCTGTCTACCAGA-MH9143270 | True | AAACCTGTCTACCAGA-MH9143270_contig_1 | True | 574 | IGH | IGHV1-69D | IGHD2-15 | IGHJ5 | IGHM | True | True | CARDRRSAYCSGGSCWGGDWFDPW | TGTGCGAGAGATCGTCGTTCAGCTTATTGTAGTGGTGGTAGCTGCT... | 564 | 42 | clonotype10 | clonotype10_consensus_1 | COVID-002 | NaN |
| 3 | AAACCTGTCTACCAGA-MH9143270 | True | AAACCTGTCTACCAGA-MH9143270_contig_2 | True | 744 | IGL | IGLV1-51 | None | IGLJ3 | IGLC3 | True | False | CGTWDSSLSAWVF | TGCGGAACATGGGATAGCAGCCTGAGTGCTTGGGTGTTC | 188 | 21 | clonotype10 | None | COVID-002 | NaN |
| 4 | AAACGGGCATTTGCTT-MH9143270 | True | AAACGGGCATTTGCTT-MH9143270_contig_1 | True | 565 | IGH | IGHV3-33 | None | IGHJ4 | IGHM | True | True | CANPTRPYSSSWWYFDYW | TGTGCGAACCCCACCCGTCCATATAGCAGCAGCTGGTGGTACTTTG... | 234 | 20 | clonotype11 | clonotype11_consensus_1 | COVID-002 | NaN |
The Table contains the following entries, that will be relevant for our preprocessing or analysis. A more detailed explanation of Cell Ranger specific entries can be found here (https://support.10xgenomics.com/single-cell-vdj/software/pipelines/latest/output/annotation). In other data formats entries (e.g. AIRR) will have similar, deviating names, however the underlying information remains similar.
barcode: tag of the cell the contig was measured from
is_cell: indicates whether the barcode is associated with a cell
high_confidence: confidence of the measurement being a IR
chain: chain of the IR (e.g. TRA: T Cell Receptor α-chain, IGH: Immunoglobulin Heavy chain)
{v,d,j,c}_gene: gene used to form the specific segment of the IR
full_length: whether the full IR was captured (see below)
productive: whether the IR is productive (see below)
cdr3{_nt}: {Nucleotide} sequence of the CDR3 of the IR chain
patient_id: ID indicating the different patients
40.7.2. Productive AIRs#
Even though we can detect the AIR sequence, it might not be productive, i.e., it might not form a valid AIR. Sequences, which do not result in functional AIRs, are therefore flagged as non-productive. These are usually ignored, when loading data by tools such as Scirpy, and not used for any downstream analysis. Productive Immune receptors are defined by 10x Genomics here as:
Sequences spanning over from a V gene to a J-gene
Having a start codon in the leading region
Containing a CDR3 in the frame of the start codon.
Do not contain a stop codon within the V-J span
Example 1: Sequences not spanning over a full IR (V to J gene) as indicated by the full_length column. You can find missing gene annotation here especially for V and J genes.
columns = [
"barcode",
"v_gene",
"d_gene",
"c_gene",
"j_gene",
"productive",
"full_length",
]
df_bcr_raw[~df_bcr_raw["full_length"]][columns].head()
| barcode | v_gene | d_gene | c_gene | j_gene | productive | full_length | |
|---|---|---|---|---|---|---|---|
| 34 | AACTCCCCAAGTACCT-MH9143270 | None | None | IGLC3 | IGLJ5 | False | False |
| 38 | AACTCTTCAGATCGGA-MH9143270 | IGKV1-39 | None | IGKC | None | False | False |
| 43 | AACTGGTCAGGGAGAG-MH9143270 | IGLV4-3 | None | IGLC2 | None | False | False |
| 65 | ACACCAAGTCGATTGT-MH9143270 | None | IGHD5-12 | IGHD | IGHJ4 | False | False |
| 76 | ACATCAGCATACTACG-MH9143270 | None | IGHD3-3 | IGHD | IGHJ6 | False | False |
Example 2: Contigs express full length but there is not identifiable CDR3.
columns += ["cdr3", "cdr3_nt"]
df_bcr_raw[(df_bcr_raw["productive"] == "False") & (df_bcr_raw["full_length"])][
columns
].head(5)
| barcode | v_gene | d_gene | c_gene | j_gene | productive | full_length | cdr3 | cdr3_nt | |
|---|---|---|---|---|---|---|---|---|---|
| 3 | AAACCTGTCTACCAGA-MH9143270 | IGLV1-51 | None | IGLC3 | IGLJ3 | False | True | CGTWDSSLSAWVF | TGCGGAACATGGGATAGCAGCCTGAGTGCTTGGGTGTTC |
| 13 | AAAGCAAGTCGAAAGC-MH9143270 | IGHV5-51 | IGHD2-15 | IGHD | IGHJ4 | False | True | None | None |
| 23 | AACGTTGAGCTGCGAA-MH9143270 | IGHV3-30 | None | IGHM | IGHJ4 | False | True | CESGRATITMIVVTDLDYW | TGCGAAAGCGGGCGGGCTACGATTACTATGATAGTAGTGACTGACC... |
| 27 | AACTCAGTCAAAGTAG-MH9143270 | IGHV3-23 | IGHD3-16 | IGHD | IGHJ4 | False | True | CAKAWIGLDSEIYYDYIWGSYRIDFDYW | TGTGCGAAAGCTTGGATCGGACTCGACTCCGAGATTTATTATGATT... |
| 30 | AACTCCCAGGTGCACA-MH9143270 | IGKV6-21 | None | IGKC | IGKJ3 | False | True | None | None |
We will now load the TCR data and convert it from tab-separated format to comma-separated format for later use. Since barcodes overlap between multiple samples within the dataset, we further copy the unique CellID as barcode annotation. This data also contains annotation on patient-level (e.g. age, outcome, …) and cell-level annotation (cell type) in addition to the contig annotations previously discussed.
df_tcr_raw = pd.read_csv(path_tcr_input, sep="\t")
df_tcr_raw["barcode"] = df_tcr_raw.pop("CellID")
df_tcr_raw.to_csv(path_tcr_csv)
print(f"Total measurements: {len(df_tcr_raw)}")
df_tcr_raw.head()
Total measurements: 547630
| Centre | barcode | is_cell | contig_id | high_confidence | length | chain | v_gene | d_gene | j_gene | ... | Days_from_onset | time_after_LPS | Worst_Clinical_Status | Outcome | leiden | consensus | initial_clustering | study_id | AgeRange | Age | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 | Newcastle | AAACCTGAGAAACCTA-MH9179824 | True | AAACCTGAGAAACCTA-1_contig_2 | True | 512 | TRB | TRBV5-1 | None | TRBJ2-3 | ... | 8 | NaN | Moderate | Home | 26 | CD8 | CD8 | COVID-014 | (50, 59] | 54 |
| 1 | Newcastle | AAACCTGAGAAACCTA-MH9179824 | True | AAACCTGAGAAACCTA-1_contig_1 | True | 573 | TRA | TRAV8-4 | None | TRAJ31 | ... | 8 | NaN | Moderate | Home | 26 | CD8 | CD8 | COVID-014 | (50, 59] | 54 |
| 2 | Newcastle | AAACCTGAGAACAATC-MH9143324 | True | AAACCTGAGAACAATC-1_contig_1 | True | 635 | TRB | TRBV19 | None | TRBJ1-5 | ... | 16 | NaN | Moderate | Home | 25 | gdT | gdT | COVID-029 | (20, 29] | 26 |
| 3 | Newcastle | AAACCTGAGAACTCGG-MH8919329 | True | AAACCTGAGAACTCGG-1_contig_1 | True | 499 | TRA | TRAV34 | None | TRAJ30 | ... | 5 | NaN | Death | Death | 23 | Treg | Treg | COVID-045 | (70, 79] | 77 |
| 4 | Newcastle | AAACCTGAGAACTCGG-MH8919329 | True | AAACCTGAGAACTCGG-1_contig_2 | True | 496 | TRB | TRBV5-1 | None | TRBJ2-5 | ... | 5 | NaN | Death | Death | 23 | Treg | Treg | COVID-045 | (70, 79] | 77 |
5 rows × 47 columns
We will find that this data already has been processed: All contigs are of full length and represent a productive IR.
df_tcr_raw["full_length"].value_counts()
True 547630
Name: full_length, dtype: int64
df_tcr_raw["productive"].value_counts()
True 547630
Name: productive, dtype: int64
40.8. Cell-Aligned Data#
In order to perform analysis on cell level, we next need to align IR information previously sorted by contig to a cell based format via the cell barcode. Note that usually many contigs map to the same cell, since every IR consists of VJ and VDJ chain. Also, it has been shown, that a cell can express multiple IRs [Schuldt and Binstadt, 2019].
We will utilize the Python package Scirpy, that performs the cell-alignment automatically, when reading various TCR information from various formats:
adata_tcr = ir.io.read_10x_vdj(path_tcr_csv)
print(f"Amount cells: {len(adata_tcr)}")
WARNING: Non-standard locus name ignored: Multi
Amount cells: 280045
The IR information will be stored sorted in adata_tcr.obs as a Pandas DataFrame with the index representing an individual cell. The DataFrame contains the following columns:
has_ir: indicates whether an IR was detected
multi_chain: indicates whether more than 2 IRs were detected
extra_chains: list of additional chains if multi_chain is true
The contig data we observed in the raw data is stored as well. It is stored as separate columns for the two chains (VJ and VDJ) with up to 2 contigs per chain for each cell:
IR_V{D}J_{1,2}_locus
IR_V{D}J_{1,2}_productive
IR_V{D}J_{1,2}_{v,d,j,c}_call
IR_V{D}J_{1,2}_junction{_aa}
adata_tcr.obs.head(5)
| is_cell | high_confidence | multi_chain | extra_chains | IR_VJ_1_c_call | IR_VJ_2_c_call | IR_VDJ_1_c_call | IR_VDJ_2_c_call | IR_VJ_1_consensus_count | IR_VJ_2_consensus_count | ... | IR_VDJ_2_locus | IR_VJ_1_productive | IR_VJ_2_productive | IR_VDJ_1_productive | IR_VDJ_2_productive | IR_VJ_1_v_call | IR_VJ_2_v_call | IR_VDJ_1_v_call | IR_VDJ_2_v_call | has_ir | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| cell_id | |||||||||||||||||||||
| AAACCTGAGAAACCTA-MH9179824 | True | True | False | [] | TRAC | NaN | TRBC2 | NaN | 960.0 | NaN | ... | NaN | True | None | True | None | TRAV8-4 | NaN | TRBV5-1 | NaN | True |
| AAACCTGAGAACAATC-MH9143324 | True | True | False | [] | NaN | NaN | TRBC1 | NaN | NaN | NaN | ... | NaN | None | None | True | None | NaN | NaN | TRBV19 | NaN | True |
| AAACCTGAGAACTCGG-MH8919329 | True | True | False | [] | TRAC | NaN | TRBC2 | NaN | 5122.0 | NaN | ... | NaN | True | None | True | None | TRAV34 | NaN | TRBV5-1 | NaN | True |
| AAACCTGAGAAGCCCA-MH9143373 | True | True | False | [] | TRAC | NaN | NaN | NaN | 25914.0 | NaN | ... | NaN | True | None | None | None | TRAV16 | NaN | NaN | NaN | True |
| AAACCTGAGAAGGACA-MH9143274 | True | True | False | [] | TRAC | NaN | TRBC2 | NaN | 3212.0 | NaN | ... | NaN | True | None | True | None | TRAV8-1 | NaN | TRBV7-6 | NaN | True |
5 rows × 45 columns
Notice, that the patient-level information is not automatically added here. Let’s add them by loading the raw data, aligning them on a cell level and indexing them by their barcode.
patient_information = [
"barcode",
"Centre",
"Sample",
"patient_id",
"Collection_Day",
"Sex",
"Swab_result",
"Status",
"Smoker",
"Status_on_day_collection",
"Status_on_day_collection_summary",
"Days_from_onset",
"time_after_LPS",
"Worst_Clinical_Status",
"Outcome",
"initial_clustering",
"study_id",
"AgeRange",
"Age",
]
df_patient = df_tcr_raw[patient_information].copy()
df_patient["Days_from_onset"] = df_patient["Days_from_onset"].astype(
str
) # mixed type (str, int)
df_patient = df_patient.drop_duplicates().reset_index(drop=True)
# Assigning barcode as index
df_patient.index = df_patient.pop("barcode")
df_patient.index.name = None
df_patient.head()
| Centre | Sample | patient_id | Collection_Day | Sex | Swab_result | Status | Smoker | Status_on_day_collection | Status_on_day_collection_summary | Days_from_onset | time_after_LPS | Worst_Clinical_Status | Outcome | initial_clustering | study_id | AgeRange | Age | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AAACCTGAGAAACCTA-MH9179824 | Newcastle | MH9179824 | COVID-014 | D0 | Female | Positive | Covid | Non-smoker | Ward_O2 | Moderate | 8 | NaN | Moderate | Home | CD8 | COVID-014 | (50, 59] | 54 |
| AAACCTGAGAACAATC-MH9143324 | Newcastle | MH9143324 | COVID-029 | D0 | Male | Positive | Covid | Non-smoker | Ward_O2 | Moderate | 16 | NaN | Moderate | Home | gdT | COVID-029 | (20, 29] | 26 |
| AAACCTGAGAACTCGG-MH8919329 | Newcastle | MH8919329 | COVID-045 | D0 | Female | Positive | Covid | Non-smoker | ITU_intubated | Critical | 5 | NaN | Death | Death | Treg | COVID-045 | (70, 79] | 77 |
| AAACCTGAGAAGCCCA-MH9143373 | Newcastle | MH9143373 | COVID-060 | D0 | Male | Negative | Non_covid | Smoker | Ward_O2 | Non_covid | 3 | NaN | Non-covid | Home | CD4 | COVID-060 | (50, 59] | 54 |
| AAACCTGAGAAGGACA-MH9143274 | Newcastle | MH9143274 | COVID-037 | D0 | Female | Positive | Covid | Non-smoker | ITU_O2 | Severe | 21 | NaN | Severe | Home | CD4 | COVID-037 | (50, 59] | 59 |
We can now add this information to the adata object and check, whether the annotation is added as observation.
adata_tcr.obs[df_patient.columns] = df_patient
adata_tcr.obs.head()
| is_cell | high_confidence | multi_chain | extra_chains | IR_VJ_1_c_call | IR_VJ_2_c_call | IR_VDJ_1_c_call | IR_VDJ_2_c_call | IR_VJ_1_consensus_count | IR_VJ_2_consensus_count | ... | Status_on_day_collection | Status_on_day_collection_summary | Days_from_onset | time_after_LPS | Worst_Clinical_Status | Outcome | initial_clustering | study_id | AgeRange | Age | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| cell_id | |||||||||||||||||||||
| AAACCTGAGAAACCTA-MH9179824 | True | True | False | [] | TRAC | NaN | TRBC2 | NaN | 960.0 | NaN | ... | Ward_O2 | Moderate | 8 | NaN | Moderate | Home | CD8 | COVID-014 | (50, 59] | 54 |
| AAACCTGAGAACAATC-MH9143324 | True | True | False | [] | NaN | NaN | TRBC1 | NaN | NaN | NaN | ... | Ward_O2 | Moderate | 16 | NaN | Moderate | Home | gdT | COVID-029 | (20, 29] | 26 |
| AAACCTGAGAACTCGG-MH8919329 | True | True | False | [] | TRAC | NaN | TRBC2 | NaN | 5122.0 | NaN | ... | ITU_intubated | Critical | 5 | NaN | Death | Death | Treg | COVID-045 | (70, 79] | 77 |
| AAACCTGAGAAGCCCA-MH9143373 | True | True | False | [] | TRAC | NaN | NaN | NaN | 25914.0 | NaN | ... | Ward_O2 | Non_covid | 3 | NaN | Non-covid | Home | CD4 | COVID-060 | (50, 59] | 54 |
| AAACCTGAGAAGGACA-MH9143274 | True | True | False | [] | TRAC | NaN | TRBC2 | NaN | 3212.0 | NaN | ... | ITU_O2 | Severe | 21 | NaN | Severe | Home | CD4 | COVID-037 | (50, 59] | 59 |
5 rows × 63 columns
In the same manner we can load the BCR data and check the IR annotation.
adata_bcr = ir.io.read_10x_vdj(path_bcr_input)
adata_bcr.obs.head(5)
WARNING: Non-standard locus name ignored: Multi
WARNING: Non-standard locus name ignored: None
| is_cell | high_confidence | multi_chain | extra_chains | IR_VJ_1_c_call | IR_VJ_2_c_call | IR_VDJ_1_c_call | IR_VDJ_2_c_call | IR_VJ_1_consensus_count | IR_VJ_2_consensus_count | ... | IR_VDJ_2_locus | IR_VJ_1_productive | IR_VJ_2_productive | IR_VDJ_1_productive | IR_VDJ_2_productive | IR_VJ_1_v_call | IR_VJ_2_v_call | IR_VDJ_1_v_call | IR_VDJ_2_v_call | has_ir | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| cell_id | |||||||||||||||||||||
| AAACCTGAGAACTCGG-MH9143420 | True | True | False | [] | IGKC | NaN | IGHM | NaN | 6982.0 | NaN | ... | NaN | True | None | True | None | IGKV3-11 | NaN | IGHV3-23 | NaN | True |
| AAACCTGAGAATAGGG-MH8919227 | True | True | False | [] | IGKC | NaN | IGHA1 | NaN | 626.0 | NaN | ... | NaN | True | None | True | None | IGKV3-15 | NaN | IGHV1-24 | NaN | True |
| AAACCTGAGAATGTGT-MH8919326 | True | True | False | [{"c_call": "IGLC1", "consensus_count": 748, "... | IGKC | NaN | IGHM | NaN | 1028.0 | NaN | ... | NaN | True | None | True | None | IGKV1-33 | NaN | IGHV1-2 | NaN | True |
| AAACCTGAGAATGTGT-MH9143424 | True | True | False | [] | IGLC2 | NaN | NaN | NaN | 736.0 | NaN | ... | NaN | True | None | None | None | IGLV3-21 | NaN | NaN | NaN | True |
| AAACCTGAGACACTAA-MH9143423 | True | True | False | [] | IGKC | NaN | IGHG1 | NaN | 66.0 | NaN | ... | NaN | True | None | True | None | IGKV3-20 | NaN | IGHV3-23 | NaN | True |
5 rows × 45 columns
As before, we can add extract additional information annotated for each contig.
patient_information = ["barcode", "patient_id"]
df_patient = df_bcr_raw[patient_information].copy()
df_patient = df_patient.drop_duplicates().reset_index(drop=True)
# Assigning barcode as index
df_patient.index = df_patient.pop("barcode")
df_patient.index.name = None
df_patient.head()
| patient_id | |
|---|---|
| AAACCTGAGCGATATA-MH9143270 | COVID-002 |
| AAACCTGTCTACCAGA-MH9143270 | COVID-002 |
| AAACGGGCATTTGCTT-MH9143270 | COVID-002 |
| AAACGGGGTAGCACGA-MH9143270 | COVID-002 |
| AAACGGGTCGTACCGG-MH9143270 | COVID-002 |
And add them to the cell annotations:
adata_bcr.obs[df_patient.columns] = df_patient
adata_bcr.obs.head()
| is_cell | high_confidence | multi_chain | extra_chains | IR_VJ_1_c_call | IR_VJ_2_c_call | IR_VDJ_1_c_call | IR_VDJ_2_c_call | IR_VJ_1_consensus_count | IR_VJ_2_consensus_count | ... | IR_VJ_1_productive | IR_VJ_2_productive | IR_VDJ_1_productive | IR_VDJ_2_productive | IR_VJ_1_v_call | IR_VJ_2_v_call | IR_VDJ_1_v_call | IR_VDJ_2_v_call | has_ir | patient_id | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| cell_id | |||||||||||||||||||||
| AAACCTGAGAACTCGG-MH9143420 | True | True | False | [] | IGKC | NaN | IGHM | NaN | 6982.0 | NaN | ... | True | None | True | None | IGKV3-11 | NaN | IGHV3-23 | NaN | True | COVID-011 |
| AAACCTGAGAATAGGG-MH8919227 | True | True | False | [] | IGKC | NaN | IGHA1 | NaN | 626.0 | NaN | ... | True | None | True | None | IGKV3-15 | NaN | IGHV1-24 | NaN | True | COVID-075 |
| AAACCTGAGAATGTGT-MH8919326 | True | True | False | [{"c_call": "IGLC1", "consensus_count": 748, "... | IGKC | NaN | IGHM | NaN | 1028.0 | NaN | ... | True | None | True | None | IGKV1-33 | NaN | IGHV1-2 | NaN | True | COVID-003 |
| AAACCTGAGAATGTGT-MH9143424 | True | True | False | [] | IGLC2 | NaN | NaN | NaN | 736.0 | NaN | ... | True | None | None | None | IGLV3-21 | NaN | NaN | NaN | True | COVID-039 |
| AAACCTGAGACACTAA-MH9143423 | True | True | False | [] | IGKC | NaN | IGHG1 | NaN | 66.0 | NaN | ... | True | None | True | None | IGKV3-20 | NaN | IGHV3-23 | NaN | True | COVID-036 |
5 rows × 46 columns
40.9. Quality Control#
For analysis, we rely on high quality input data. It is therefore of great importance, to identify cells with incorrect or incomplete AIR information:
Incomplete AIRs: A cell is assigned only either a VJ and or a VDJ chain, because the other chain is missed during sequencing. While these are still valid cells, we cannot utilize them for downstream analysis, when full AIR sequence information is required.
Multiple AIRs: It is also possible that a cell gets assigned multiple AIRs. While it has been observed that T- / B-cells can express dual AIR [Schuldt and Binstadt, 2019], cells with more than two IRs are indicative as doublets, which should not be used for downstream analysis.
We can easily assign this state of the AIR by accumulating the data of the AIR chains. This will enable us later to filter cells, where the AIR cannot be used for our analysis. Here, it heavily depends on your downstream analysis which AIR information is required.
40.9.1. Assigning AIR State#
ir.tl.chain_qc(adata_tcr)
ir.tl.chain_qc(adata_bcr)
The following provides the resulting column with an explanation of its possible options:
chain_pairing
orphan {VJ}/{VDJ}: cells with only one of VJ or VDJ chain
single pair: cells with a full paired (VJ and VDJ) AIR
extra {VJ}/{VDJ}: cells with full paired AIR and an additional VJ or VDJ chain
two full chains: cells with two paired AIRs
multichain: cells with more than two VJ or VDJ chains (likely doublets)
receptor_type: BCR, TCR, no IR, or ambiguous (mixed chains of TCR and BCR)
receptor_subtype: α/β, ɣ/δ, IG-λ, and IG-κ chain configurations
40.9.2. Visualization#
Depending on the data, the quality of the chain pairing might be different. You should visualize the results over different conditions (e.g. samples, data sources, …), that could introduce varying quality within your dataset. Let’s visualize the data quality collected over the different centers of data collection.
_ = ir.pl.group_abundance(adata_tcr, groupby="Centre", target_col="chain_pairing")
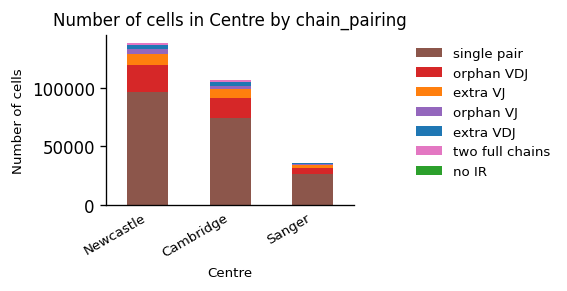
The data from all three centers contains mainly cells with a single pair of TCRs, which is a sign of good data quality. Often, we can compare different conditions better, when we view the fractions of cells in each condition instead of the absolute amount of cells:
_ = ir.pl.group_abundance(
adata_tcr, groupby="Centre", target_col="chain_pairing", normalize=True
)
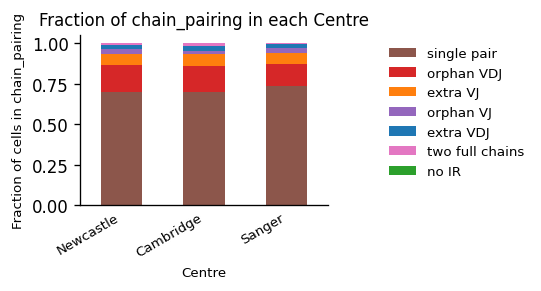
Here, we can see that all centers offer data of overall comparable data quality. The majority of T cells express a single TCR, followed by orphan VDJ, and VJ chains, which is typical when sequencing TCRs. Typical values of a good quality sample may be greater 60% single AIRs, 10-20% orphan chains, and ~10% extra chains. Note, that the percentage of cells without AIRs is naturally depending on the amount of non-AIR cells in the study, when sequencing multiple-modalities jointly.
_ = ir.pl.group_abundance(
adata_tcr, groupby="Centre", target_col="receptor_type", normalize=True
)
_ = ir.pl.group_abundance(
adata_tcr, groupby="Centre", target_col="receptor_subtype", normalize=True
)


We can see, that the dataset consists only of TCRs (first plot), which is not surprising, since we have separated DataFrames of T - and B-cells. Additionally, the dataset only contains α/β-T cells. Since the amount of cells without IR cannot be recognized in the plot, we will look at the absolute numbers, and observe that only 22 cells have no AIR information. This fraction will be larger in most datasets, when sequencing multiple modalities depending on the amount of AIR cells.
adata_tcr.obs["chain_pairing"].value_counts()
single pair 196957
orphan VDJ 45266
extra VJ 19034
orphan VJ 7937
extra VDJ 7473
two full chains 3356
no IR 22
Name: chain_pairing, dtype: int64
For the BCR, we will have a look at the patient-level. For visualization, we downsample to two patients:
adata_bcr_tmp = adata_bcr[
adata_bcr.obs["patient_id"].isin(["COVID-003", "AP11"])
].copy()
_ = ir.pl.group_abundance(
adata_bcr_tmp, groupby="patient_id", target_col="chain_pairing"
)
_ = ir.pl.group_abundance(
adata_bcr_tmp, groupby="patient_id", target_col="chain_pairing", normalize=True
)
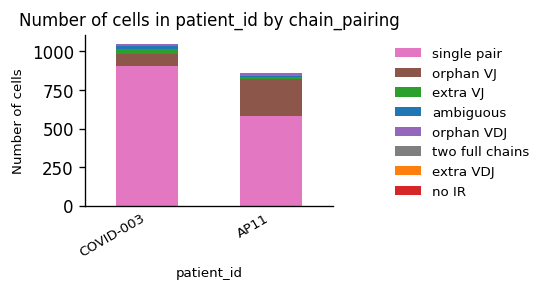
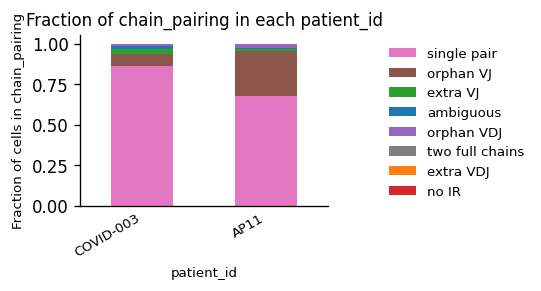
While we observe an considerable difference in the amount of B Cells between the patients, the quality of the chain pairings is comparable.
_ = ir.pl.group_abundance(
adata_bcr_tmp, groupby="patient_id", target_col="receptor_type", normalize=True
)
_ = ir.pl.group_abundance(
adata_bcr_tmp, groupby="patient_id", target_col="receptor_subtype", normalize=True
)

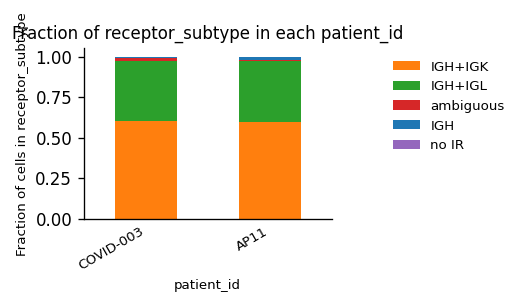
Again, we have only one type of AIR in the dataset (BCRs), but this time a mixture of κ- and λ-chains for the light chain.
40.9.3. Filtering#
You might want to filter cells with different AIR states. Depending on your study, you have to decide the trade-off between the amount of data available and the quality of the data, when deciding for filtering. Often, all cells are kept within the dataset with their AIR state flagged. For different downstream analysis, only cells with correct information are then used. E.g. if you perform a database query based on CDR3-VDJ chain (see chapter X), it is possible to use orphan VDJ chains, while this is not possible when searching for the full AIR. In the following, we will show how the filtering is performed. It is up to the reader to decide, whether they want to perform the filtering at this point, or before the different analysis parts.
The filtering can be performed with various degrees of strictness and different combinations such as:
has IR: keep only cells with expressed IRs, since we cannot perform sequence level analysis on them
no multi chains: filter out cells with >2 AIRs to avoid doublets
only one AIR: filter out cells with two AIRs or additional chains since specificity can not be attributed to a certain sequence
no single chains: filter out cells with only VJ or VDJ chains, which cannot be used for all downstream analysis due to missing information
Since we will use the full data throughout this book, we will filter the data to a temporary AnnData object for each step described above. From the absolute cell numbers, you can see how many cells will be lost in each step.
adata_bcr_tmp.obs["chain_pairing"].value_counts()
single pair 1489
orphan VJ 312
extra VJ 46
ambiguous 31
orphan VDJ 25
two full chains 6
extra VDJ 4
no IR 1
Name: chain_pairing, dtype: int64
print(f"Amount of all B cells:\t\t\t\t{len(adata_bcr)}")
adata_bcr_tmp = adata_bcr[adata_bcr.obs["chain_pairing"] != "no IR"]
print(f"Amount of B cells with AIR:\t\t\t{len(adata_bcr_tmp)}")
adata_bcr_tmp = adata_bcr_tmp[adata_bcr_tmp.obs["chain_pairing"] != "multi_chain"]
print(f"Amount of B cells without doublets:\t\t{len(adata_bcr_tmp)}")
adata_bcr_tmp = adata_bcr_tmp[
~adata_bcr_tmp.obs["chain_pairing"].isin(
["two full chains", "extra VJ", "extra VDJ"]
)
]
print(f"Amount of B cells with unique AIR per cell:\t{len(adata_bcr_tmp)}")
adata_bcr_tmp = adata_bcr_tmp[adata_bcr_tmp.obs["chain_pairing"] == "single pair"]
print(f"Amount of B cells with sinlge complete AIR:\t{len(adata_bcr_tmp)}")
Amount of all B cells: 159446
Amount of B cells with AIR: 159185
Amount of B cells without doublets: 159185
Amount of B cells with unique AIR per cell: 153936
Amount of B cells with single complete AIR: 108395
Eventually, we obtain an AnnData object with only single paired BCRs. However, the amount of data can be considerably smaller limiting downstream analysis.
adata_bcr_tmp.obs["chain_pairing"].value_counts()
single pair 108395
Name: chain_pairing, dtype: int64
Similarly, we can perform this filtering for the TCR data.
adata_tcr.obs["chain_pairing"].value_counts()
single pair 196957
orphan VDJ 45266
extra VJ 19034
orphan VJ 7937
extra VDJ 7473
two full chains 3356
no IR 22
Name: chain_pairing, dtype: int64
print(f"Amount of all T cells:\t\t\t\t{len(adata_tcr)}")
adata_tcr_tmp = adata_tcr[adata_tcr.obs["chain_pairing"] != "no IR"]
print(f"Amount of T cells with AIR:\t\t\t{len(adata_tcr_tmp)}")
adata_tcr_tmp = adata_tcr_tmp[adata_tcr_tmp.obs["chain_pairing"] != "multi_chain"]
print(f"Amount of T cells without doublets:\t\t{len(adata_tcr_tmp)}")
adata_tcr_tmp = adata_tcr_tmp[
~adata_tcr_tmp.obs["chain_pairing"].isin(
["two full chains", "extra VJ", "extra VDJ"]
)
]
print(f"Amount of T cells with unique AIR per cell:\t{len(adata_tcr_tmp)}")
adata_tcr_tmp = adata_tcr_tmp[adata_tcr_tmp.obs["chain_pairing"] == "single pair"]
print(f"Amount of T cells with sinlge complete AIR:\t{len(adata_tcr_tmp)}")
Amount of all T cells: 280045
Amount of T cells with AIR: 280023
Amount of T cells without doublets: 280023
Amount of T cells with unique AIR per cell: 250160
Amount of T cells with sinlge complete AIR: 196957
adata_tcr_tmp.obs["chain_pairing"].value_counts()
single pair 196957
Name: chain_pairing, dtype: int64
Finally, we save the annotated data for later analysis.
sc.write(adata=adata_tcr, filename=path_tcr_out)
sc.write(adata=adata_bcr, filename=path_bcr_out)
40.10. Quiz#
40.11. References#
Nicholas Borcherding, Nicholas L Bormann, and Gloria Kraus. Screpertoire: an r-based toolkit for single-cell immune receptor analysis. F1000Research, 2020.
Bryan Briney, Anne Inderbitzin, Collin Joyce, and Dennis R Burton. Commonality despite exceptional diversity in the baseline human antibody repertoire. Nature, 566(7744):393–397, 2019.
Stefan Canzar, Karlynn E Neu, Qingming Tang, Patrick C Wilson, and Aly A Khan. BASIC: BCR assembly from single cells. Bioinformatics, 33(3):425–427, 10 2016. URL: https://doi.org/10.1093/bioinformatics/btw631, arXiv:https://academic.oup.com/bioinformatics/article-pdf/33/3/425/25145999/btw631.pdf, doi:10.1093/bioinformatics/btw631.
Max D Cooper and Matthew N Alder. The evolution of adaptive immune systems. Cell, 124(4):815–822, 2006.
Ping Hu, Wenhua Zhang, Hongbo Xin, and Glenn Deng. Single cell isolation and analysis. Frontiers in cell and developmental biology, 4:116, 2016.
Ida Lindeman, Guy Emerton, Lira Mamanova, Omri Snir, Krzysztof Polanski, Shuo-Wang Qiao, Ludvig M. Sollid, Sarah A. Teichmann, and Michael J. T. Stubbington. Bracer: b-cell-receptor reconstruction and clonality inference from single-cell rna-seq. Nature Methods, 15(8):563–565, Aug 2018. URL: https://doi.org/10.1038/s41592-018-0082-3, doi:10.1038/s41592-018-0082-3.
Nathaniel J Schuldt and Bryce A Binstadt. Dual tcr t cells: identity crisis or multitaskers? The Journal of Immunology, 202(3):637–644, 2019.
Mandeep Singh, Ghamdan Al-Eryani, Shaun Carswell, James M Ferguson, James Blackburn, Kirston Barton, Daniel Roden, Fabio Luciani, Tri Giang Phan, Simon Junankar, and others. High-throughput targeted long-read single cell sequencing reveals the clonal and transcriptional landscape of lymphocytes. Nature communications, 10(1):1–13, 2019.
Emily Stephenson, Gary Reynolds, Rachel A Botting, Fernando J Calero-Nieto, Michael D Morgan, Zewen Kelvin Tuong, Karsten Bach, Waradon Sungnak, Kaylee B Worlock, Masahiro Yoshida, and others. Single-cell multi-omics analysis of the immune response in covid-19. Nature medicine, 27(5):904–916, 2021.
Gregor Sturm, Tamas Szabo, Georgios Fotakis, Marlene Haider, Dietmar Rieder, Zlatko Trajanoski, and Francesca Finotello. Scirpy: a scanpy extension for analyzing single-cell t-cell receptor-sequencing data. Bioinformatics, 36(18):4817–4818, 2020.
Amit A. Upadhyay, Robert C. Kauffman, Amber N. Wolabaugh, Alice Cho, Nirav B. Patel, Samantha M. Reiss, Colin Havenar-Daughton, Reem A. Dawoud, Gregory K. Tharp, Iñaki Sanz, Bali Pulendran, Shane Crotty, F. Eun-Hyung Lee, Jens Wrammert, and Steven E. Bosinger. Baldr: a computational pipeline for paired heavy and light chain immunoglobulin reconstruction in single-cell rna-seq data. Genome Medicine, 10(1):20, Mar 2018. URL: https://doi.org/10.1186/s13073-018-0528-3, doi:10.1186/s13073-018-0528-3.
Sebastiaan Valkiers, Nicky de Vrij, Sofie Gielis, Sara Verbandt, Benson Ogunjimi, Kris Laukens, and Pieter Meysman. Recent advances in t-cell receptor repertoire analysis: bridging the gap with multimodal single-cell rna sequencing. ImmunoInformatics, pages 100009, 2022.
F Alexander Wolf, Philipp Angerer, and Fabian J Theis. Scanpy: large-scale single-cell gene expression data analysis. Genome biology, 19(1):1–5, 2018.
Alexander Yermanos, Andreas Agrafiotis, Raphael Kuhn, Damiano Robbiani, Josephine Yates, Chrysa Papadopoulou, Jiami Han, Ioana Sandu, Cédric Weber, Florian Bieberich, and others. Platypus: an open-access software for integrating lymphocyte single-cell immune repertoires with transcriptomes. NAR genomics and bioinformatics, 3(2):lqab023, 2021.
Veronika Zarnitsyna, Brian Evavold, Louie Schoettle, Joseph Blattman, and Rustom Antia. Estimating the diversity, completeness, and cross-reactivity of the t cell repertoire. Frontiers in immunology, 4:485, 2013.
ImmunoMind Team. immunarch: An R Package for Painless Bioinformatics Analysis of T-Cell and B-Cell Immune Repertoires. August 2019. URL: https://doi.org/10.5281/zenodo.3367200, doi:10.5281/zenodo.3367200.
