17. Differential gene expression analysis#
Key takeaways
Repeated measurements (cells) from the same experimental subject in scRNA-seq introduce correlations between measurements, which need to be adjusted by modeling the experimental units as Random Effect to mitigate the pseudoreplication issue. Account for them through a sum or mean aggregation (pseudobulk methods - by fixed- or random effect terms) or by accounting for individual as a random effect (single-cell methods) to mitigate the pseudoreplication issue.
Statistical power is best increased with more samples in the experimental design.
Environment setup
Install conda:
Before creating the environment, ensure that conda is installed on your system.
Save the yml content:
Copy the content from the yml tab into a file named
environment.yml.
Create the environment:
Open a terminal or command prompt.
Run the following command:
conda env create -f environment.yml
Activate the environment:
After the environment is created, activate it using:
conda activate <environment_name>
Replace
<environment_name>with the name specified in theenvironment.ymlfile. In the yml file it will look like this:name: <environment_name>
Verify the installation:
Check that the environment was created successfully by running:
conda env list
name: differential-gene-expression
channels:
- conda-forge
- bioconda
- defaults
dependencies:
- conda-forge::python=3.12.12
- conda-forge::r-base=4.4.3
- bioconda::bioconductor-mast=1.32.0
- bioconda::bioconductor-edger=4.4.0
- bioconda::bioconductor-singlecellexperiment=1.28.0
- conda-forge::r-lme4
- conda-forge::rpy2=3.6.4
- bioconda::anndata2ri=2.0
- conda-forge::pip
- pip:
- pertpy==1.0.4
17.1. Motivation#
This chapter is a more detailed continuation of the Annotation subchapter which already introduced differential gene expression (DGE) as a tool to annotate clusters with cell types. Here, we focus on more advanced use-cases of differential gene expression testing on more complex experimental designs which involve one or more conditions such as diseases, genetic knockouts or drugs. In such cases we are commonly interested in the magnitude and significance of differences in gene expression patterns between the condition of interest and a reference. This reference can be everything but is commonly a healthy sample. This statistical test can be applied to arbitrary groups, but in the case of single-cell RNA-Seq is commonly applied on the cell type level.
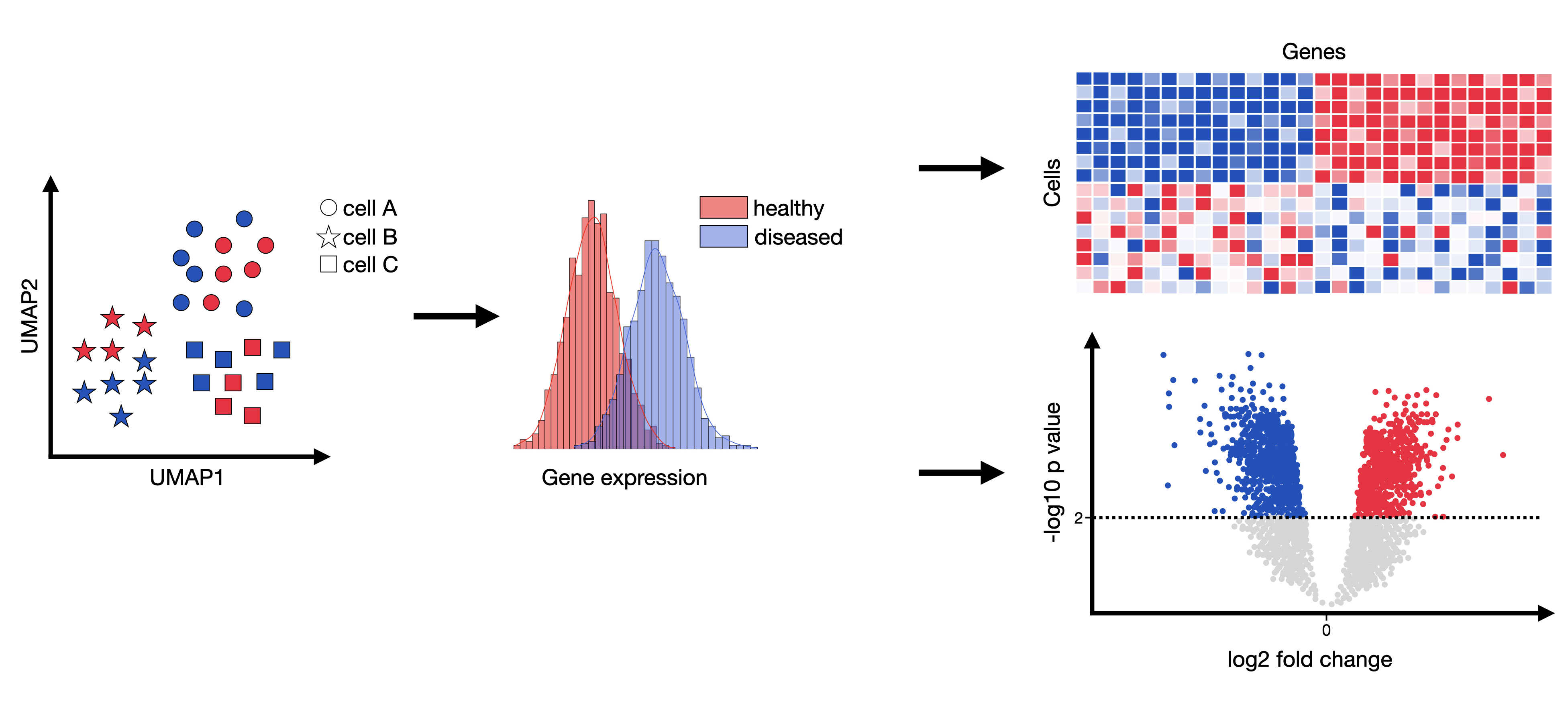
Fig. 17.1 Differential gene expression analysis attempts to infer genes that are statistically significantly over- or underexpressed between any compared groups (commonly between healthy and condition per cell type).#
The outcome of such an analysis could be genesets which effect and potentially explain any observed phenotypes. These genesets can then be examined more closely with respect to, for example, affected pathways or induced cell-cell communication changes.
A differential gene expression test usually returns the log2 fold-change and the adjusted p-value per compared genes per compared conditions. This list can then be sorted by p-value and investigated in more detail.
The popular student’s t-test is one way of conducting such a test. However, it fails to take several single-cell RNA-seq peculiarities into account such as the excess number of zeros originating from dropouts or the need for complex experimental designs. More specifically, very rarely does one have sufficient sample numbers to accurately estimate the variance without pooling information across genes. Moreover, raw counts are never an absolute measurement of expression for a specific gene within a given sample. The actual read number per gene depends on the efficiency of the library preparation, the amount of contamination from non-coding transcripts and the sequencing depth. It does therefore lack in both, sensitivity and specificity for single-cell RNA-seq, let alone experimental design flexibility.
As a result, differential gene expression testing is a classic bioinformatics problem which has been tackled by many tools already. Generally, the problem is currently being approached from two views, the sample-level view where expression is aggregated to create “pseudobulks” and then analysed with methods originally designed for bulk expression samples such as edgeR[Robinson et al., 2010] or DEseq2[Love et al., 2014] and the cell-level view where cells are modeled individually using generalized mixed effect models such as MAST[Finak et al., 2015] or glmmTMB[Brooks et al., 2017]. The consensus and robustness across datasets for DGE tools is low [Das et al., 2021, Wang et al., 2019]. As previously described, although single-cell data contains technical noise artifacts such as dropout, zero-inflation and high cell-to-cell variability [Hicks et al., 2017, Luecken and Theis, 2019, Vallejos et al., 2017], methods designed for bulk RNA-seq data performed favorably compared to methods explicitly designed for scRNA-seq data[Das et al., 2021, Jaakkola et al., 2016, Soneson and Robinson, 2018, Squair et al., 2021]. Single-cell specific methods were found to be especially prone to wrongly labeling highly expressed genes as differentially expressed.
A recent study highlighted the issue of pseudoreplication where inferential statistics is applied to biological replicates which are not statistically independent. Failing to account for the inherent correlation of replicates (cells from the same individual) inflates the false discovery rate (FDR)[Junttila et al., 2022, Squair et al., 2021, Zimmerman et al., 2021]. Therefore, batch effect correction or the aggregation of cell-type-specific expression values within an individual through either a sum, mean or random effect per individual, that is pseudobulk generation, should be applied prior to DGE analysis to account for within-sample correlations [Zimmerman et al., 2021]. Generally, both, pseudobulk methods with sum aggregation such as edgeR, DESeq2, or Limma[Ritchie et al., 2015] and mixed models such as MAST with random effect setting were found to be superior compared to naive methods, such as the popular Wilcoxon rank-sum test or Seurat’s [Hao et al., 2021] latent models, which do not account for them[Junttila et al., 2022].
In matters arising from the Zimmerman paper, Murphy et al. critically examined the Zimmerman benchmarking strategy and improved it [Murphy and Skene, 2022]. They came to the conclusion that pseudobulk methods perform best but whether sum or mean aggregation works better requires further investigation.
Hence, in this notebook, we demonstrate how to use two tools for DE analysis: edgeR with a quasi-likelihood test and MAST with random effects. Since both edgeR and MAST are implemented in R, we use the anndata2ri package to be able to simultaneously work with AnnData objects in Python and SingeCellExperiment objects in R.
For each of the two methods, we show two use cases: how to run the analysis on full data and perform testing on multiple cell types and on one specific cell type.
17.2. Environment setup#
import warnings
warnings.filterwarnings("ignore")
import logging
import random
import anndata2ri
import matplotlib.pyplot as plt
import numpy as np
import pandas as pd
import pertpy
import rpy2.rinterface_lib.callbacks
import sc_toolbox
import scanpy as sc
import seaborn as sns
from rpy2.robjects import pandas2ri
sc.settings.verbosity = 0
rpy2.rinterface_lib.callbacks.logger.setLevel(logging.ERROR)
pandas2ri.activate()
anndata2ri.activate()
%load_ext rpy2.ipython
To use sccoda or tasccoda please install ete3 with pip install ete3
%%R
library(edgeR)
library(MAST)
17.3. Preparing the dataset#
We will use the Kang dataset, which is a 10x droplet-based scRNA-seq peripheral blood mononuclear cell (PBMC) data from 8 Lupus patients before and after 6h-treatment with INF-β (16 samples in total)[Kang et al., 2018]. Interferon beta is used in the form of natural fibroblast or recombinant preparations (interferon beta-1a and interferon beta-1b) and exerts antiviral and antiproliferative properties similar to those of interferon alpha. Interferon beta has been approved for the treatment of relapsing–remitting multiple sclerosis and secondary progressive multiple sclerosis.
First, we load the full dataset.
adata = pertpy.data.kang_2018()
adata
AnnData object with n_obs × n_vars = 24673 × 15706
obs: 'nCount_RNA', 'nFeature_RNA', 'tsne1', 'tsne2', 'label', 'cluster', 'cell_type', 'replicate', 'nCount_SCT', 'nFeature_SCT', 'integrated_snn_res.0.4', 'seurat_clusters'
var: 'name'
obsm: 'X_pca', 'X_umap'
We will need label (which contains the condition label), replicate and cell_type columns of the .obs.
adata.obs[:5]
| nCount_RNA | nFeature_RNA | tsne1 | tsne2 | label | cluster | cell_type | replicate | nCount_SCT | nFeature_SCT | integrated_snn_res.0.4 | seurat_clusters | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| index | ||||||||||||
| AAACATACATTTCC-1 | 3017.0 | 877 | -27.640373 | 14.966629 | ctrl | 9 | CD14+ Monocytes | patient_1016 | 1704.0 | 711 | 1 | 1 |
| AAACATACCAGAAA-1 | 2481.0 | 713 | -27.493646 | 28.924885 | ctrl | 9 | CD14+ Monocytes | patient_1256 | 1614.0 | 662 | 1 | 1 |
| AAACATACCATGCA-1 | 703.0 | 337 | -10.468194 | -5.984389 | ctrl | 3 | CD4 T cells | patient_1488 | 908.0 | 337 | 6 | 6 |
| AAACATACCTCGCT-1 | 3420.0 | 850 | -24.367997 | 20.429285 | ctrl | 9 | CD14+ Monocytes | patient_1256 | 1738.0 | 653 | 1 | 1 |
| AAACATACCTGGTA-1 | 3158.0 | 1111 | 27.952170 | 24.159738 | ctrl | 4 | Dendritic cells | patient_1039 | 1857.0 | 928 | 12 | 12 |
We will need to work with raw counts so we check that .X indeed contains raw counts and put them into the counts layer of our AnnData object.
np.max(adata.X)
3828.0
adata.layers["counts"] = adata.X.copy()
We have 8 control and 8 disease patients.
print(len(adata[adata.obs["label"] == "ctrl"].obs["replicate"].cat.categories))
print(len(adata[adata.obs["label"] == "stim"].obs["replicate"].cat.categories))
8
8
We filter cells which have less than 200 genes and genes which were found in less than 3 cells for a rudimentary quality control.
sc.pp.filter_cells(adata, min_genes=200)
sc.pp.filter_genes(adata, min_cells=3)
adata
AnnData object with n_obs × n_vars = 24562 × 15701
obs: 'nCount_RNA', 'nFeature_RNA', 'tsne1', 'tsne2', 'label', 'cluster', 'cell_type', 'replicate', 'nCount_SCT', 'nFeature_SCT', 'integrated_snn_res.0.4', 'seurat_clusters', 'n_genes'
var: 'name', 'n_cells'
obsm: 'X_pca', 'X_umap'
layers: 'counts'
17.4. Pseudobulk#
edgeR is a differential gene expression testing tool implemented in R which was initially designed for bulk gene expression data. It implements a wide range of statistical methodologies based on the negative binomial distribution, empirical Bayes estimation, exact tests, generalized linear models (GLMs) and quasi-likelihood tests.
For more details please read the original publication[Robinson et al., 2010].
Here, we will be using the quasi-likelihood test because it accounts for the uncertainty of the dispersion estimates. In contrast, the exact test assumes that the estimated dispersion is the true value, which can result in some inaccuracy. Additionally, the quasi-likelihood GLMs are more flexible when it comes to experimental design.
Since edgeR was introduced as a method for DE analysis for bulk data, we first need to create pseudobulk samples from our single-cell dataset. For each patient we create 1 pseudobulk sample per cell type by aggregating the cell from each subpopulation and taking the mean gene expression within that subpopulation.
If the data you are working with does not have replicates, it could be beneficial to create multiple (e.g. 2-3) pseudobulks per patient to account for patient variability. Here we chose to create 1 pseudobulk per patient because for each patient we have 2 replicates, one control and one stimulated.
We strongly recommend to read this guide https://f1000research.com/articles/9-1444 on design matrices.
Regardless of whether we want to run the analysis only on a few cell subpopulations and fit a model for each one of them separately or fit one model for all of them, we first need to prepare the data, define a function to create pseudobulks and run the edgeR pipeline. First, let’s prepare the data.
Since we need to create pseudobulks for each patient-condition combination, we first need to create such a column by concatenating replicate and label.
adata.obs["sample"] = [
f"{rep}_{l}" for rep, l in zip(adata.obs["replicate"], adata.obs["label"], strict=False)
]
We need to clean up the cell type names, i.e. replace spaces with underscores and remove plus-signs, to avoid Python to R conversion issues.
adata.obs["cell_type"] = [ct.replace(" ", "_") for ct in adata.obs["cell_type"]]
adata.obs["cell_type"] = [ct.replace("+", "") for ct in adata.obs["cell_type"]]
We need to set categorical metadata to be indeed categorical to create pseudobulks.
adata.obs["replicate"] = adata.obs["replicate"].astype("category")
adata.obs["label"] = adata.obs["label"].astype("category")
adata.obs["sample"] = adata.obs["sample"].astype("category")
adata.obs["cell_type"] = adata.obs["cell_type"].astype("category")
Now, let’s define the function we need to aggregate single cells into pseudo-replicates:
aggregate_and_filteris a function that creates an AnnData object with one pseudo-replicate for each donor for a specified subpopulation from the original single-cell AnnData object. Here, we also filter out donors that have fewer than 30 cells for the specified population.by changing the
replicates_per_patientparameter, several (n) pseudo-replicates can be created for each sample; cells are then split into n subsets of roughly equal sizes.
NUM_OF_CELL_PER_DONOR = 30
def aggregate_and_filter(
adata,
cell_identity,
donor_key="sample",
condition_key="label",
cell_identity_key="cell_type",
obs_to_keep=None, # which additional metadata to keep, e.g. gender, age, etc.
replicates_per_patient=1,
):
# subset adata to the given cell identity
if obs_to_keep is None:
obs_to_keep = []
adata_cell_pop = adata[adata.obs[cell_identity_key] == cell_identity].copy()
# check which donors to keep according to the number of cells specified with NUM_OF_CELL_PER_DONOR
size_by_donor = adata_cell_pop.obs.groupby([donor_key]).size()
donors_to_drop = [
donor
for donor in size_by_donor.index
if size_by_donor[donor] <= NUM_OF_CELL_PER_DONOR
]
if len(donors_to_drop) > 0:
print("Dropping the following samples:")
print(donors_to_drop)
df = pd.DataFrame(columns=[*adata_cell_pop.var_names, *obs_to_keep])
adata_cell_pop.obs[donor_key] = adata_cell_pop.obs[donor_key].astype("category")
for i, donor in enumerate(donors := adata_cell_pop.obs[donor_key].cat.categories):
print(f"\tProcessing donor {i+1} out of {len(donors)}...", end="\r")
if donor not in donors_to_drop:
adata_donor = adata_cell_pop[adata_cell_pop.obs[donor_key] == donor]
# create replicates for each donor
indices = list(adata_donor.obs_names)
random.shuffle(indices)
indices = np.array_split(np.array(indices), replicates_per_patient)
for i, rep_idx in enumerate(indices):
adata_replicate = adata_donor[rep_idx]
# specify how to aggregate: sum gene expression for each gene for each donor and also keep the condition information
agg_dict = dict.fromkeys(adata_replicate.var_names, "sum")
for obs in obs_to_keep:
agg_dict[obs] = "first"
# create a df with all genes, donor and condition info
df_donor = pd.DataFrame(adata_replicate.X.A)
df_donor.index = adata_replicate.obs_names
df_donor.columns = adata_replicate.var_names
df_donor = df_donor.join(adata_replicate.obs[obs_to_keep])
# aggregate
df_donor = df_donor.groupby(donor_key).agg(agg_dict)
df_donor[donor_key] = donor
df.loc[f"donor_{donor}_{i}"] = df_donor.loc[donor]
print("\n")
# create AnnData object from the df
adata_cell_pop = sc.AnnData(
df[adata_cell_pop.var_names], obs=df.drop(columns=adata_cell_pop.var_names)
)
return adata_cell_pop
We also need to define a separate function to fit an edgeR GLM:
fit_modeltakes a SingleCellExperiment object as input, creates the design matrix and outputs the fitted GLM. We also output the edgeR object of class DGEList to do some exploratory data analysis (EDA).
%%R
fit_model <- function(adata_){
# create an edgeR object with counts and grouping factor
y <- DGEList(assay(adata_, "X"), group = colData(adata_)$label)
# filter out genes with low counts
print("Dimensions before subsetting:")
print(dim(y))
print("")
keep <- filterByExpr(y)
y <- y[keep, , keep.lib.sizes=FALSE]
print("Dimensions after subsetting:")
print(dim(y))
print("")
# normalize
y <- calcNormFactors(y)
# create a vector that is a concatenation of condition and cell type that we will later use with contrasts
group <- paste0(colData(adata_)$label, ".", colData(adata_)$cell_type)
replicate <- colData(adata_)$replicate
# create a design matrix: here we have multiple donors so also consider that in the design matrix
design <- model.matrix(~ 0 + group + replicate)
# estimate dispersion
y <- estimateDisp(y, design = design)
# fit the model
fit <- glmQLFit(y, design)
return(list("fit"=fit, "design"=design, "y"=y))
}
Now we have defined all the functions we need, so we can proceed with creating pseudobulks. We might want to look at available metadata later and therefore keep it in the AnnData object.
obs_to_keep = ["label", "cell_type", "replicate", "sample"]
We need to pass the raw counts to edgeR. Hence, we set .X to the counts layer to ensure the pseudo-replicates are created for raw counts.
adata.X = adata.layers["counts"].copy()
Next, we create the AnnData object with pseudobulks.
# process first cell type separately...
cell_type = adata.obs["cell_type"].cat.categories[0]
print(
f'Processing {cell_type} (1 out of {len(adata.obs["cell_type"].cat.categories)})...'
)
adata_pb = aggregate_and_filter(adata, cell_type, obs_to_keep=obs_to_keep)
for i, cell_type in enumerate(adata.obs["cell_type"].cat.categories[1:]):
print(
f'Processing {cell_type} ({i+2} out of {len(adata.obs["cell_type"].cat.categories)})...'
)
adata_cell_type = aggregate_and_filter(adata, cell_type, obs_to_keep=obs_to_keep)
adata_pb = adata_pb.concatenate(adata_cell_type)
Processing B_cells (1 out of 8)...
Dropping the following samples:
['patient_1039_ctrl']
Processing donor 16 out of 16...
Processing CD14_Monocytes (2 out of 8)...
Processing donor 16 out of 16...
Processing CD4_T_cells (3 out of 8)...
Processing donor 16 out of 16...
Processing CD8_T_cells (4 out of 8)...
Dropping the following samples:
['patient_101_ctrl', 'patient_1039_ctrl', 'patient_1039_stim', 'patient_107_ctrl', 'patient_107_stim', 'patient_1244_ctrl', 'patient_1244_stim']
Processing donor 16 out of 16...
Processing Dendritic_cells (5 out of 8)...
Dropping the following samples:
['patient_1016_ctrl', 'patient_1016_stim', 'patient_101_ctrl', 'patient_1039_ctrl', 'patient_1039_stim', 'patient_107_ctrl', 'patient_107_stim']
Processing donor 16 out of 16...
Processing FCGR3A_Monocytes (6 out of 8)...
Dropping the following samples:
['patient_1039_ctrl', 'patient_1039_stim', 'patient_107_ctrl', 'patient_107_stim', 'patient_1244_stim']
Processing donor 16 out of 16...
Processing Megakaryocytes (7 out of 8)...
Dropping the following samples:
['patient_1015_ctrl', 'patient_1015_stim', 'patient_1016_ctrl', 'patient_101_ctrl', 'patient_1039_stim', 'patient_107_stim', 'patient_1244_ctrl', 'patient_1244_stim', 'patient_1256_ctrl', 'patient_1256_stim', 'patient_1488_ctrl']
Processing donor 11 out of 11...
Processing NK_cells (8 out of 8)...
Dropping the following samples:
['patient_1039_ctrl', 'patient_1039_stim']
Processing donor 16 out of 16...
The validity of differential gene expression results highly depends on the capture of the major axis of variations in the statistical model. Intermediate data exploration steps such as principal component analysis (PCA) or multidimensional scaling (MDS) on pseudobulk samples allow for the identification of the sources of variation and thus can guide the construction of corresponding design and contrast matrices that model the data[Law et al., 2020].
Failing to account for multiple sources of biological variability for experiments which include biological replicates will inflate the FDR[Thurman et al., 2021],[Lähnemann et al., 2020]. While increasing the number of cells per individual increases the precision, it has a limited effect on the power for the detection of differences across individuals. Therefore, the best way to increase statistical power is to increase the number of independent experimental samples[Zimmerman et al., 2021].
Since our data has already been generated, we cannot further increase the number of independent experimental samples. Nevertheless, we will now explore our data to determine the major axes of variation to properly generate our design matrices.
We perform very basic EDA on the created pseudo-replicates to check if some patients/pseudobulks are outliers that we need to exclude so as not to bias the DE results. We save the raw counts in the 'counts' layer, then normalize the counts and calculate the PCA coordinates for the normalized pseudobulk counts.
adata_pb.layers['counts'] = adata_pb.X.copy()
sc.pp.normalize_total(adata_pb, target_sum=1e6)
sc.pp.log1p(adata_pb)
sc.pp.pca(adata_pb)
Next, we look at created pseudo-replicates on a PCA plot and color by all the available metadata to see if there are any confounding factors that we might want to include in the design matrix. We also add a lib_size and log_lib_size columns to check if there is a correlation between library size and PC components.
adata_pb.obs["lib_size"] = np.sum(adata_pb.layers["counts"], axis=1)
adata_pb.obs["log_lib_size"] = np.log(adata_pb.obs["lib_size"])
sc.pl.pca(adata_pb, color=adata_pb.obs, ncols=1, size=300)
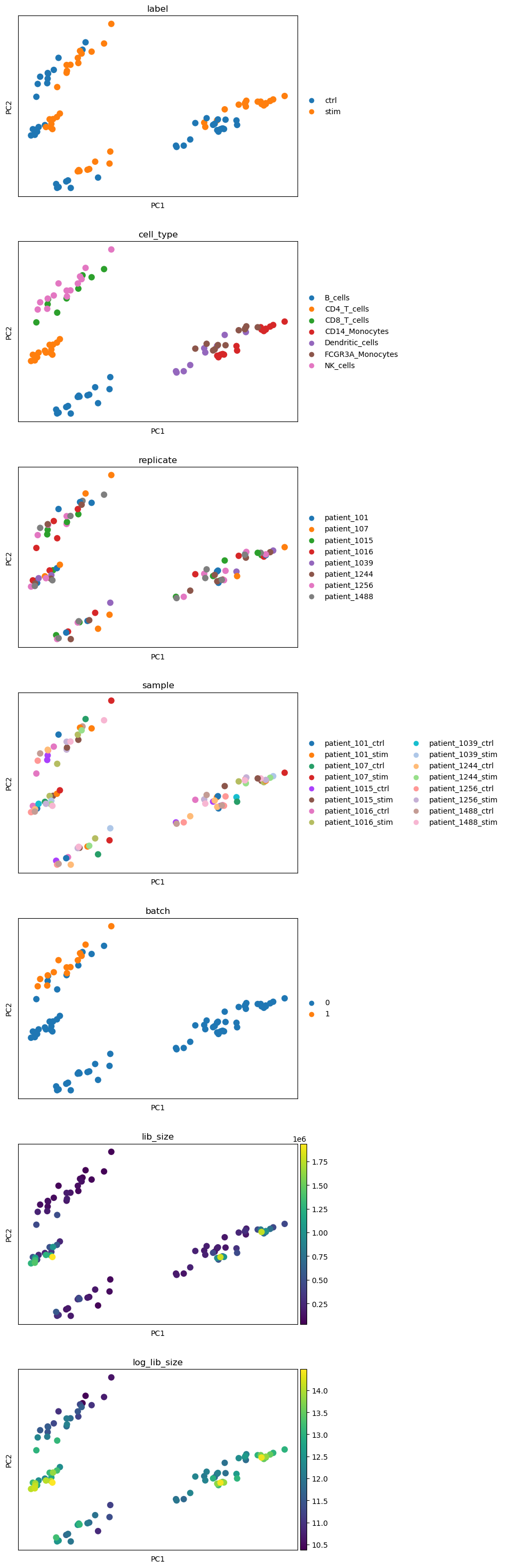
We observe separation of cell types on the PCA plots as well as the separation into stimulated and unstimulated cells. Other covariates (batch) do not seem to be clearly correlated with the PCA components so we do not include any of them in our design matrix.
As mentioned above, edgeR takes raw counts as input, so we put counts back into the .X field before we proceed.
adata_pb.X = adata_pb.layers['counts'].copy()
17.4.1. One group#
First, we show how to prepare the data, construct the design matrix and perform the DE testing for one specific cell type.
We run the pipeline on CD14+ Monocytes subset of the data, as it was shown in the paper that the highest number of DE genes was identified in this subpopulation.
adata_mono = adata_pb[adata_pb.obs["cell_type"] == "CD14_Monocytes"]
adata_mono
View of AnnData object with n_obs × n_vars = 16 × 15701
obs: 'label', 'cell_type', 'replicate', 'sample', 'batch', 'lib_size', 'log_lib_size'
uns: 'log1p', 'pca', 'label_colors', 'cell_type_colors', 'replicate_colors', 'sample_colors', 'batch_colors'
obsm: 'X_pca'
varm: 'PCs'
layers: 'counts'
Clean the sample names to make plots less crowded.
adata_mono.obs_names = [
name.split("_")[2] + "_" + name.split("_")[3] for name in adata_mono.obs_names
]
%%time
%%R -i adata_mono
outs <-fit_model(adata_mono)
[1] "Dimensions before subsetting:"
[1] 15701 16
[1] ""
[1] "Dimensions after subsetting:"
[1] 3709 16
[1] ""
CPU times: user 2.95 s, sys: 224 ms, total: 3.18 s
Wall time: 4.27 s
%%R
fit <- outs$fit
y <- outs$y
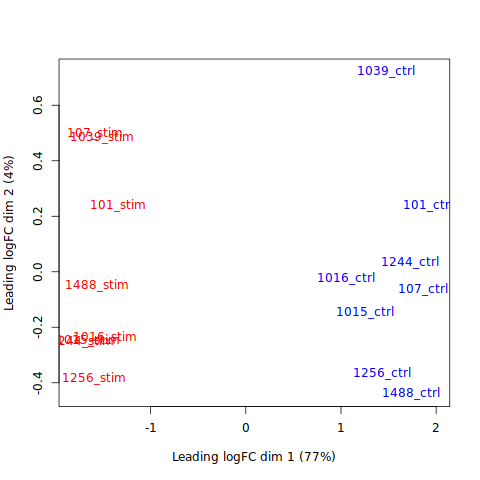
Since we did not enter our analysis with a prior assumption that a specific gene will be up or downregulated, we need visualizations to make sense of the DGE results. MDS plots allow for a high level overview. Commonly, we expect a separation between samples from different conditions as can be seen in the following plot for our results.
%%R
plotMDS(y, col=ifelse(y$samples$group == "stim", "red", "blue"))
Fontconfig warning: ignoring UTF-8: not a valid region tag
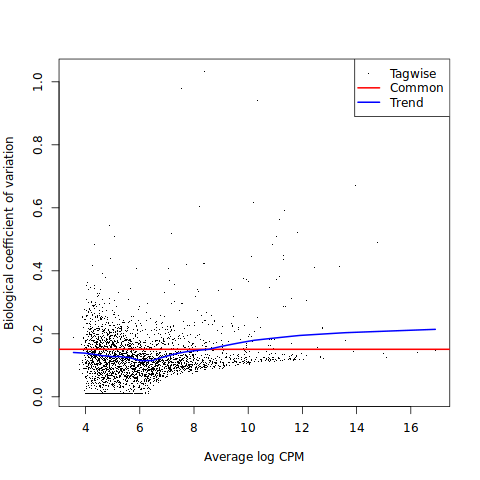
Biological Coefficient of Variation (BCV) plots show the variability of every gene on average across biological groups as a function of mean expression. For example, bcv of 0.3 indicates that there is on average 30% percent variability in the expression of genes across groups.
Genes with low abundance generally exhibit larger BCV as read count measurements are more uncertain for low abundance genes. On the other hand, genes with high average expression are quantified more reliably, thus they generally have lower variability and hence lower BCV. Use this plot to detect outlier genes or pinpoint other experimental factors that may need to be reflected in the design matrix. For example, a group of genes with high average expression and high BCV appearing in the top right corner of the plot can flag experimental stress, contamination etc, particularly if they belong to similar gene families.
BCV is the square root of dispersion. The distance between tagwise and common bcv trend indicates if tagwise (gene-wise) dispersion estimates are highly variable (i.e. heterogenous gene expression). If tag-wise dispersion values are very heterogeneous, less moderation is applied to capture heterogeneity. The distance between any two points on those curves reflects how much the dispersion for a gene was shrunken towards the common trend. That is, it captures the amount of dispersion moderation applied.
In the BCV plot below, we see some low abundance genes with high bcv, but no high abundance genes with high bcv, which indicates that there should not be any further experimental considerations modelled into the design matrix.
%%R
plotBCV(y)
Next, we perform the quasi-likelihood test to find DE genes between control and stimulated conditions. Let’s check what columns our design matrix has to specify the correct columns to test on.
%%R
colnames(y$design)
[1] "groupctrl.CD14_Monocytes" "groupstim.CD14_Monocytes"
[3] "replicatepatient_107" "replicatepatient_1015"
[5] "replicatepatient_1016" "replicatepatient_1039"
[7] "replicatepatient_1244" "replicatepatient_1256"
[9] "replicatepatient_1488"
%%R -o tt
myContrast <- makeContrasts('groupstim.CD14_Monocytes-groupctrl.CD14_Monocytes', levels = y$design)
qlf <- glmQLFTest(fit, contrast=myContrast)
# get all of the DE genes and calculate Benjamini-Hochberg adjusted FDR
tt <- topTags(qlf, n = Inf)
tt <- tt$table
Let’s inspect the table: for each of the genes that were not filtered out by edgeR (3709 in our case), the table contains the results of the DE testing. The table can be saved as a .csv file to work with later and used for visualization. We will show how to use volcano plots at the end of the section after we run the analysis on full data.
tt.shape
(3709, 5)
tt[:5]
| logFC | logCPM | F | PValue | FDR | |
|---|---|---|---|---|---|
| HESX1 | 8.345536 | 6.773420 | 1281.013295 | 1.837373e-15 | 2.766927e-12 |
| CD38 | 7.126846 | 7.420668 | 1243.793133 | 2.266164e-15 | 2.766927e-12 |
| NT5C3A | 5.657050 | 8.327003 | 1218.102628 | 2.628780e-15 | 2.766927e-12 |
| SOCS1 | 4.388247 | 6.943768 | 1191.289806 | 3.079524e-15 | 2.766927e-12 |
| GMPR | 6.943484 | 7.031832 | 1159.601183 | 3.730018e-15 | 2.766927e-12 |
As a side note, one can also test for genes that are differentially expressed for a provided coefficient or contrast relative to a specified fold-change using glmTreat.
%%R
tr <- glmTreat(fit, contrast=myContrast, lfc=1.5)
print(head(topTags(tr)))
Coefficient: -1*groupctrl.CD14_Monocytes 1*groupstim.CD14_Monocytes
logFC unshrunk.logFC logCPM PValue FDR
HESX1 8.345536 8.702486 6.773420 2.108737e-14 3.535749e-11
CD38 7.126846 7.219184 7.420668 2.292459e-14 3.535749e-11
NT5C3A 5.657050 5.674640 8.327003 3.149544e-14 3.535749e-11
IL1RN 6.588583 6.596787 10.358946 4.249159e-14 3.535749e-11
GMPR 6.943484 7.047504 7.031832 4.766446e-14 3.535749e-11
DEFB1 6.654201 6.696759 8.067159 7.670147e-14 4.741429e-11
And finally, we can see how many genes we have with FDR-corrected values of less than 0.01.
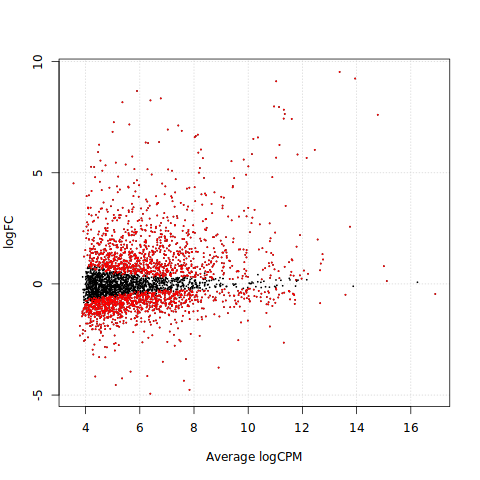
The smear plot (also known as MA, M-values vs A-values, plot) shows the log fold-change of the genes as a function of their mean abundance. We generally observe higher logFC at low abundance ranges as read counts are more variable at low abundance resulting in large logFC estimates. If we fit a loess curve to logFC and Average logCPM values, the trend should center around zero. Any deviations from this can indicate that data has not been properly normalised. Genes with large mean expression and large logFC in absolute values can flag biologically interesting genes for investigation and follow-up.
%%R
plotSmear(qlf, de.tags = rownames(tt)[which(tt$FDR<0.01)])
17.4.2. Multiple groups#
Next, we show how to prepare the data, construct the design matrix and perform the DE testing using contrasts on the full dataset for all available cell types.
%%time
%%R -i adata_pb
outs <-fit_model(adata_pb)
[1] "Dimensions before subsetting:"
[1] 15701 90
[1] ""
[1] "Dimensions after subsetting:"
[1] 2358 90
[1] ""
CPU times: user 11.9 s, sys: 39.7 ms, total: 12 s
Wall time: 12 s
%%R
fit <- outs$fit
y <- outs$y
Now we use contrasts to perform a quasi-likelihood test for each of our cell types. Because there is no straightforward way to move tables from R to Python from within an R loop we get the results manually for each cell type.
%%R -i adata_pb -o de_per_cell_type
de_per_cell_type <- list()
for (cell_type in unique(colData(adata_pb)$cell_type)) {
print(cell_type)
# create contrast for this cell type
myContrast <- makeContrasts(paste0("groupstim.", cell_type, "-groupctrl.", cell_type), levels = y$design)
# perform QLF test
qlf <- glmQLFTest(fit, contrast=myContrast)
# get all of the DE genes and calculate Benjamini-Hochberg adjusted FDR
tt <- topTags(qlf, n = Inf)
# save in the list with the results for all the cell types
de_per_cell_type[[cell_type]] <- tt$table
}
[1] "B_cells"
[1] "CD14_Monocytes"
[1] "CD4_T_cells"
[1] "CD8_T_cells"
[1] "Dendritic_cells"
[1] "FCGR3A_Monocytes"
[1] "NK_cells"
Now let’s and save it in .uns of our original adata object using sc_toolbox.tools.de_res_to_anndata from sc_toolbox package (schillerlab/sc-toolbox), which saves the results as if they were created with the scanpy’s rank_genes_groups() function. The advantage of storing the DE tables like this is that we could now also use standard scanpy plotting functions. We save the DEG tables as .csv for each cell type as we will need them later in the cell-to-cell communication chapter.
# get cell types that we ran the analysis for
cell_types = de_per_cell_type.keys()
# add the table to .uns for each cell type
for cell_type in cell_types:
df = de_per_cell_type[cell_type]
df["gene_symbol"] = df.index
df["cell_type"] = cell_type
sc_toolbox.tools.de_res_to_anndata(
adata,
df,
groupby="cell_type",
score_col="logCPM",
pval_col="PValue",
pval_adj_col="FDR",
lfc_col="logFC",
key_added="edgeR_" + cell_type,
)
df.to_csv(f"de_edgeR_{cell_type}.csv")
To get the table as a pandas DataFrame again, we use sc.get.rank_genes_groups_df function.
sc.get.rank_genes_groups_df(adata, group="CD14_Monocytes", key="edgeR_CD14_Monocytes")[
:5
]
| names | scores | logfoldchanges | pvals | pvals_adj | |
|---|---|---|---|---|---|
| 0 | FTH1 | 16.186098 | -0.572272 | 0.001589 | 0.002712 |
| 1 | MALAT1 | 16.025682 | 0.010049 | 0.877078 | 0.897245 |
| 2 | B2M | 15.468524 | 0.677266 | 0.0 | 0.0 |
| 3 | TMSB4X | 15.054697 | -0.217131 | 0.004663 | 0.007331 |
| 4 | FTL | 14.984937 | -0.041832 | 0.669951 | 0.710956 |
17.4.3. Notes on edgeR#
Requires raw counts as input
Requires pseudobulks from a single-cell experiment
If there are several donors in the single-cell experiment and the user wants to account for the patient variability, we recommend creating 2 or 3 pseudo-replicates for each patient and including patient information into the design matrix
17.5. Single-cell specific#
The MAST framework models single-cell gene expression using a two-part generalized linear model. One component of MAST models the discrete expression rate of each gene across cells, while the other component models the conditional continuous expression level (conditional on the gene being expressed). For more details please read the original publication[Finak et al., 2015].
MAST takes normalized counts as input, so we first take the ‘counts’ layer and then perform the normalization step.
adata.X = adata.layers["counts"].copy()
sc.pp.normalize_total(adata, target_sum=1e6)
sc.pp.log1p(adata)
We define a small helper function that takes care of some object type conversion issue between R and Python. We also need to filter out genes that are expressed in a small number of cells (3 in this case) for each subpopulation as the model needs to be able to estimate the variance for each gene.
def prep_anndata(adata_):
def fix_dtypes(adata_):
df = pd.DataFrame(adata_.X.A, index=adata_.obs_names, columns=adata_.var_names)
df = df.join(adata_.obs)
return sc.AnnData(df[adata_.var_names], obs=df.drop(columns=adata_.var_names))
adata_ = fix_dtypes(adata_)
sc.pp.filter_genes(adata_, min_cells=3)
return adata_
17.5.1. One group#
As with edgeR, it is possible to fit the model using the full dataset, and then use contrasts to perform the testing for each cell type of interest, but in this notebook we only show the MAST-RE pipeline for one cell type, namely CD14 Monocytes, to shorten the runtime.
adata_mono = adata[adata.obs["cell_type"] == "CD14_Monocytes"].copy()
adata_mono
AnnData object with n_obs × n_vars = 5696 × 15701
obs: 'nCount_RNA', 'nFeature_RNA', 'tsne1', 'tsne2', 'label', 'cluster', 'cell_type', 'replicate', 'nCount_SCT', 'nFeature_SCT', 'integrated_snn_res.0.4', 'seurat_clusters', 'n_genes', 'sample'
var: 'name', 'n_cells'
uns: 'edgeR_B_cells', 'edgeR_CD14_Monocytes', 'edgeR_CD4_T_cells', 'edgeR_CD8_T_cells', 'edgeR_Dendritic_cells', 'edgeR_FCGR3A_Monocytes', 'edgeR_NK_cells', 'log1p'
obsm: 'X_pca', 'X_umap'
layers: 'counts'
sc.pp.filter_genes(adata_mono, min_cells=3)
adata_mono
AnnData object with n_obs × n_vars = 5696 × 12268
obs: 'nCount_RNA', 'nFeature_RNA', 'tsne1', 'tsne2', 'label', 'cluster', 'cell_type', 'replicate', 'nCount_SCT', 'nFeature_SCT', 'integrated_snn_res.0.4', 'seurat_clusters', 'n_genes', 'sample'
var: 'name', 'n_cells'
uns: 'edgeR_B_cells', 'edgeR_CD14_Monocytes', 'edgeR_CD4_T_cells', 'edgeR_CD8_T_cells', 'edgeR_Dendritic_cells', 'edgeR_FCGR3A_Monocytes', 'edgeR_NK_cells', 'log1p'
obsm: 'X_pca', 'X_umap'
layers: 'counts'
Next we filter both objects as mentioned above.
adata_mono = prep_anndata(adata_mono)
adata_mono
AnnData object with n_obs × n_vars = 5696 × 12268
obs: 'nCount_RNA', 'nFeature_RNA', 'tsne1', 'tsne2', 'label', 'cluster', 'cell_type', 'replicate', 'nCount_SCT', 'nFeature_SCT', 'integrated_snn_res.0.4', 'seurat_clusters', 'n_genes', 'sample'
var: 'n_cells'
As is the case whenever multiple statistical tests are performed, the obtained p-values for DGE tests over conditions must be corrected for multiple testing using, for example, a Benjamini-Hochberg correction[Luecken and Theis, 2019],[Benjamini and Hochberg, 1995].
Similarly to edgeR analysis, we define a separate function that we use for the analysis:
find_de_MAST_REtakes aSingleCellExperimentobject as input and runs MAST with RE pipeline. The output of the function is a table (pandas DataFrame in Python) which contains results of the analysis, e.g. log-fold change, p-value and FDR-corrected value for each gene.
adata_mono.obs["cell_type"] = [
ct.replace(" ", "_") for ct in adata_mono.obs["cell_type"]
]
adata_mono.obs["cell_type"] = [
ct.replace("+", "") for ct in adata_mono.obs["cell_type"]
]
%%R
find_de_MAST_RE <- function(adata_){
# create a MAST object
sca <- SceToSingleCellAssay(adata_, class = "SingleCellAssay")
print("Dimensions before subsetting:")
print(dim(sca))
print("")
# keep genes that are expressed in more than 10% of all cells
sca <- sca[freq(sca)>0.1,]
print("Dimensions after subsetting:")
print(dim(sca))
print("")
# add a column to the data which contains scaled number of genes that are expressed in each cell
cdr2 <- colSums(assay(sca)>0)
colData(sca)$ngeneson <- scale(cdr2)
# store the columns that we are interested in as factors
label <- factor(colData(sca)$label)
# set the reference level
label <- relevel(label,"ctrl")
colData(sca)$label <- label
celltype <- factor(colData(sca)$cell_type)
colData(sca)$celltype <- celltype
# same for donors (which we need to model random effects)
replicate <- factor(colData(sca)$replicate)
colData(sca)$replicate <- replicate
# create a group per condition-celltype combination
colData(sca)$group <- paste0(colData(adata_)$label, ".", colData(adata_)$cell_type)
colData(sca)$group <- factor(colData(sca)$group)
# define and fit the model
zlmCond <- zlm(formula = ~ngeneson + group + (1 | replicate),
sca=sca,
method='glmer',
ebayes=F,
strictConvergence=F,
fitArgsD=list(nAGQ = 0)) # to speed up calculations
# perform likelihood-ratio test for the condition that we are interested in
summaryCond <- summary(zlmCond, doLRT='groupstim.CD14_Monocytes')
# get the table with log-fold changes and p-values
summaryDt <- summaryCond$datatable
result <- merge(summaryDt[contrast=='groupstim.CD14_Monocytes' & component=='H',.(primerid, `Pr(>Chisq)`)], # p-values
summaryDt[contrast=='groupstim.CD14_Monocytes' & component=='logFC', .(primerid, coef)],
by='primerid') # logFC coefficients
# MAST uses natural logarithm so we convert the coefficients to log2 base to be comparable to edgeR
result[,coef:=result[,coef]/log(2)]
# do multiple testing correction
result[,FDR:=p.adjust(`Pr(>Chisq)`, 'fdr')]
result = result[result$FDR<0.01,, drop=F]
result <- stats::na.omit(as.data.frame(result))
return(result)
}
We run the pipeline for monocytes.
%%time
%%R -i adata_mono -o res
res <-find_de_MAST_RE(adata_mono)
[1] "Dimensions before subsetting:"
[1] 12268 5696
[1] ""
[1] "Dimensions after subsetting:"
[1] 1676 5696
[1] ""
CPU times: user 9min 35s, sys: 2.96 s, total: 9min 38s
Wall time: 9min 43s
Let’s take a look at the results.
res[:5]
| primerid | Pr(>Chisq) | coef | FDR | |
|---|---|---|---|---|
| 1 | AAED1 | 8.410341e-19 | 0.709836 | 1.597106e-18 |
| 2 | ABI1 | 1.401688e-05 | 0.312797 | 1.824921e-05 |
| 3 | ABRACL | 3.920183e-06 | 0.418028 | 5.201004e-06 |
| 4 | ACADVL | 2.121110e-26 | -0.751305 | 4.656979e-26 |
| 5 | ACOT9 | 3.424174e-151 | 2.921133 | 2.689503e-150 |
We store the result in .uns as before. Note that we won’t need the score column so we just pass logFC there.
res["gene_symbol"] = res["primerid"]
res["cell_type"] = "CD14_Monocytes"
sc_toolbox.tools.de_res_to_anndata(
adata,
res,
groupby="cell_type",
score_col="coef",
pval_col="Pr(>Chisq)",
pval_adj_col="FDR",
lfc_col="coef",
key_added="MAST_CD14_Monocytes",
)
adata_copy = adata.copy()
17.6. Visualization#
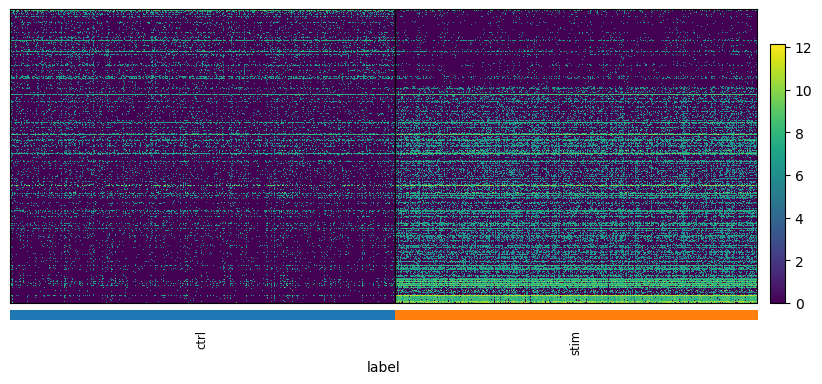
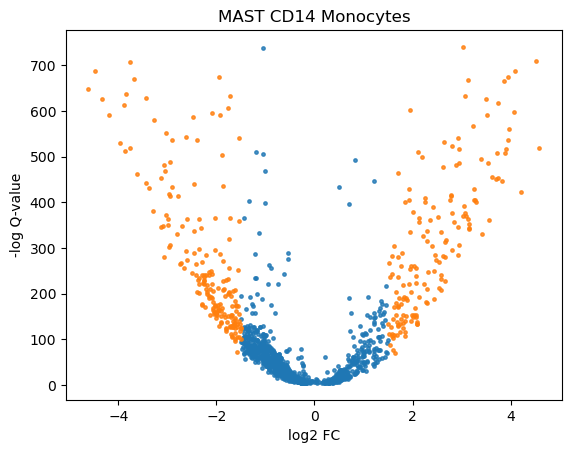
We can visualize the results with a heatmap and a volcano plot. Each row of a heatmap corresponds to a gene and each column to a single-cell. The brighter the color the higher the expression of that gene in a particular cell. Since we only plot DE genes, we would like to see clear differences in expression between the two conditions. Volcano plots are often used to visualize the results of statistical testing, and they show the change in expression on the x-axis (log-fold change) and statistical significance on the y-axis (FDR-corrected p-values). We color code the genes that have FDR-corrected p-value under 0.01 and log-fold change of over 1.5
We normalize the data before plotting the heatmaps to see the differences in expression between two conditions better.
adata.X = adata.layers["counts"].copy()
sc.pp.normalize_total(adata, target_sum=1e6)
sc.pp.log1p(adata)
Next, we define a helper plotting function for the heatmaps.
FDR = 0.01
LOG_FOLD_CHANGE = 1.5
def plot_heatmap(adata, group_key, group_name="cell_type", groupby="label"):
cell_type = "_".join(group_key.split("_")[1:])
res = sc.get.rank_genes_groups_df(adata, group=cell_type, key=group_key)
res.index = res["names"].values
res = res[
(res["pvals_adj"] < FDR) & (abs(res["logfoldchanges"]) > LOG_FOLD_CHANGE)
].sort_values(by=["logfoldchanges"])
print(f"Plotting {len(res)} genes...")
markers = list(res.index)
sc.pl.heatmap(
adata[adata.obs[group_name] == cell_type].copy(),
markers,
groupby=groupby,
swap_axes=True,
)
And finally we can plot the heatmaps with DEG for CD14+ Monocytes for both edgeR and MAST with RE.
plot_heatmap(adata, "edgeR_CD14_Monocytes")
Plotting 303 genes...
plot_heatmap(adata, "MAST_CD14_Monocytes")
Plotting 436 genes...
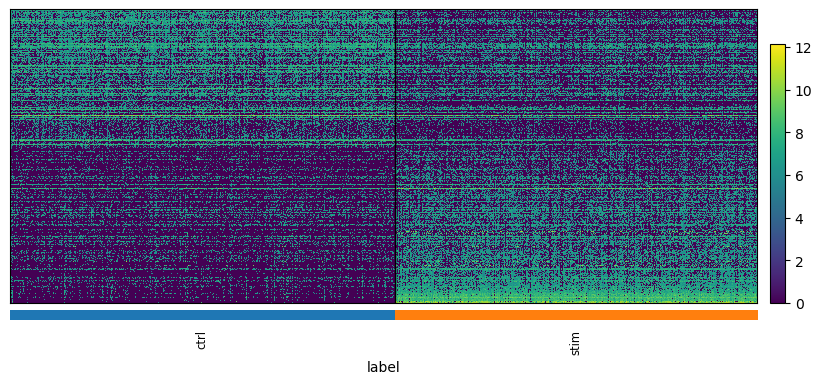
We observe that MAST identified 436 DEG with our given cut-offs for adjusted p-values and logfold change, while edgeR identified 303 genes.
Next, we define the helper plotting function for the volcano plots.
FDR = 0.01
LOG_FOLD_CHANGE = 1.5
def volcano_plot(adata, group_key, group_name="cell_type", groupby="label", title=None):
cell_type = "_".join(group_key.split("_")[1:])
result = sc.get.rank_genes_groups_df(adata, group=cell_type, key=group_key).copy()
result["-logQ"] = -np.log(result["pvals"].astype("float"))
lowqval_de = result.loc[abs(result["logfoldchanges"]) > LOG_FOLD_CHANGE]
other_de = result.loc[abs(result["logfoldchanges"]) <= LOG_FOLD_CHANGE]
fig, ax = plt.subplots()
sns.regplot(
x=other_de["logfoldchanges"],
y=other_de["-logQ"],
fit_reg=False,
scatter_kws={"s": 6},
)
sns.regplot(
x=lowqval_de["logfoldchanges"],
y=lowqval_de["-logQ"],
fit_reg=False,
scatter_kws={"s": 6},
)
ax.set_xlabel("log2 FC")
ax.set_ylabel("-log Q-value")
if title is None:
title = group_key.replace("_", " ")
plt.title(title)
plt.show()
volcano_plot(adata, "MAST_CD14_Monocytes")
volcano_plot(adata, "edgeR_CD14_Monocytes")
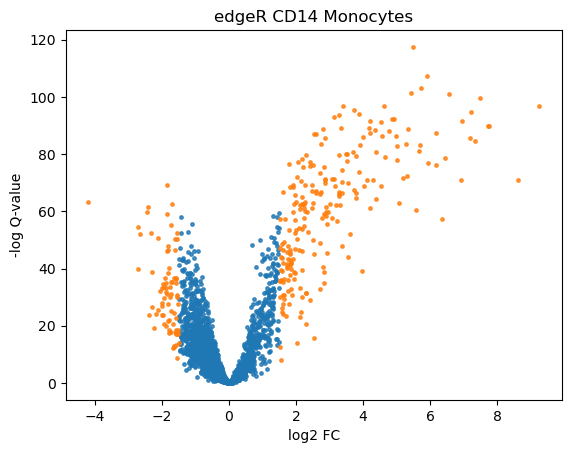
From the heatmaps and especially from the volcano plots, one can see that edgeR identified more up-regulated than down-regulated genes (in stimulated vs control) in contrast to MAST which identified similar number of up- and down-regulated genes.
17.7. Quiz#
17.8. References#
Yoav Benjamini and Yosef Hochberg. Controlling the false discovery rate: a practical and powerful approach to multiple testing. Journal of the Royal Statistical Society: Series B (Methodological), 57(1):289–300, 1995. URL: https://rss.onlinelibrary.wiley.com/doi/abs/10.1111/j.2517-6161.1995.tb02031.x, arXiv:https://rss.onlinelibrary.wiley.com/doi/pdf/10.1111/j.2517-6161.1995.tb02031.x, doi:https://doi.org/10.1111/j.2517-6161.1995.tb02031.x.
Mollie E. Brooks, Kasper Kristensen, Koen J. van Benthem, Arni Magnusson, Casper W. Berg, Anders Nielsen, Hans J. Skaug, Martin Mächler, and Benjamin M. Bolker. glmmTMB Balances Speed and Flexibility Among Packages for Zero-inflated Generalized Linear Mixed Modeling. The R Journal, 9(2):378–400, 2017. URL: https://doi.org/10.32614/RJ-2017-066, doi:10.32614/RJ-2017-066.
Samarendra Das, Anil Rai, Michael L Merchant, Matthew C Cave, and Shesh N Rai. A comprehensive survey of statistical approaches for differential expression analysis in single-cell term`RNA` sequencing studies. Genes (Basel), 12(12):1947, December 2021.
Greg Finak, Andrew McDavid, Masanao Yajima, Jingyuan Deng, Vivian Gersuk, Alex K. Shalek, Chloe K. Slichter, Hannah W. Miller, M. Juliana McElrath, Martin Prlic, Peter S. Linsley, and Raphael Gottardo. Mast: a flexible statistical framework for assessing transcriptional changes and characterizing heterogeneity in single-cell term`rna` sequencing data. Genome Biology, 16(1):278, Dec 2015. URL: https://doi.org/10.1186/s13059-015-0844-5, doi:10.1186/s13059-015-0844-5.
Yuhan Hao, Stephanie Hao, Erica Andersen-Nissen, William M. Mauck III, Shiwei Zheng, Andrew Butler, Maddie J. Lee, Aaron J. Wilk, Charlotte Darby, Michael Zagar, Paul Hoffman, Marlon Stoeckius, Efthymia Papalexi, Eleni P. Mimitou, Jaison Jain, Avi Srivastava, Tim Stuart, Lamar B. Fleming, Bertrand Yeung, Angela J. Rogers, Juliana M. McElrath, Catherine A. Blish, Raphael Gottardo, Peter Smibert, and Rahul Satija. Integrated analysis of multimodal single-cell data. Cell, 2021. URL: https://doi.org/10.1016/j.cell.2021.04.048, doi:10.1016/j.cell.2021.04.048.
Stephanie C Hicks, F William Townes, Mingxiang Teng, and Rafael A Irizarry. Missing data and technical variability in single-cell term`RNA`-sequencing experiments. Biostatistics, 19(4):562–578, 11 2017. URL: https://doi.org/10.1093/biostatistics/kxx053, arXiv:https://academic.oup.com/biostatistics/article-pdf/19/4/562/26346801/kxx053.pdf, doi:10.1093/biostatistics/kxx053.
Maria K Jaakkola, Fatemeh Seyeterm`DNA`srollah, Arfa Mehmood, and Laura L Elo. Comparison of methods to detect differentially expressed genes between single-cell populations. Briefings in Bioinformatics, 18(5):735–743, 07 2016. URL: https://doi.org/10.1093/bib/bbw057, arXiv:https://academic.oup.com/bib/article-pdf/18/5/735/25581122/bbw057.pdf, doi:10.1093/bib/bbw057.
Sini Junttila, Johannes Smolander, and Laura L Elo. Benchmarking methods for detecting differential states between conditions from multi-subject single-cell term`rna`-seq data. bioRxiv, 2022. URL: https://www.biorxiv.org/content/early/2022/02/19/2022.02.16.480662, arXiv:https://www.biorxiv.org/content/early/2022/02/19/2022.02.16.480662.full.pdf, doi:10.1101/2022.02.16.480662.
Hyun Min Kang, Meena Subramaniam, Sasha Targ, Michelle Nguyen, Lenka Maliskova, Elizabeth McCarthy, Eunice Wan, Simon Wong, Lauren Byrnes, Cristina M Lanata, and others. Multiplexed droplet single-cell term`rna`-sequencing using natural genetic variation. Nature biotechnology, 36(1):89–94, 2018.
Charity W. Law, Kathleen Zeglinski, Xueyi Dong, Monther Alhamdoosh, Gordon K. Smyth, and Matthew E. Ritchie. A guide to creating design matrices for gene expression experiments. F1000Research, 9:1444–1444, Dec 2020. 33604029[pmid]. URL: https://pubmed.ncbi.nlm.nih.gov/33604029.
Michael I. Love, Wolfgang Huber, and Simon Anders. Moderated estimation of fold change and dispersion for term`rna`-seq data with deseq2. Genome Biology, 15(12):550, Dec 2014. URL: https://doi.org/10.1186/s13059-014-0550-8, doi:10.1186/s13059-014-0550-8.
Malte D Luecken and Fabian J Theis. Current best practices in single-cell term`rna`-seq analysis: a tutorial. Molecular Systems Biology, 15(6):e8746, 2019. URL: https://www.embopress.org/doi/abs/10.15252/msb.20188746, arXiv:https://www.embopress.org/doi/pdf/10.15252/msb.20188746, doi:https://doi.org/10.15252/msb.20188746.
David Lähnemann, Johannes Köster, Ewa Szczurek, Davis J. McCarthy, Stephanie C. Hicks, Mark D. Robinson, Catalina A. Vallejos, Kieran R. Campbell, Niko Beerenwinkel, Ahmed Mahfouz, Luca Pinello, Pavel Skums, Alexandros Stamatakis, Camille Stephan-Otto Attolini, Samuel Aparicio, Jasmijn Baaijens, Marleen Balvert, Buys de Barbanson, Antonio Cappuccio, Giacomo Corleone, Bas E. Dutilh, Maria Florescu, Victor Guryev, Rens Holmer, Katharina Jahn, Thamar Jessurun Lobo, Emma M. Keizer, Indu Khatri, Szymon M. Kielbasa, Jan O. Korbel, Alexey M. Kozlov, Tzu-Hao Kuo, Boudewijn P.F. Lelieveldt, Ion I. Mandoiu, John C. Marioni, Tobias Marschall, Felix Mölder, Amir Niknejad, Lukasz Raczkowski, Marcel Reinders, Jeroen de Ridder, Antoine-Emmanuel Saliba, Antonios Somarakis, Oliver Stegle, Fabian J. Theis, Huan Yang, Alex Zelikovsky, Alice C. McHardy, Benjamin J. Raphael, Sohrab P. Shah, and Alexander Schönhuth. Eleven grand challenges in single-cell data science. Genome Biology, 21(1):31, Feb 2020. URL: https://doi.org/10.1186/s13059-020-1926-6, doi:10.1186/s13059-020-1926-6.
Alan E. Murphy and Nathan G. Skene. A balanced measure shows superior performance of pseudobulk methods in single-cell RNA-sequencing analysis. Nat Commun, dec 2022. URL: https://doi.org/10.1038%2Fs41467-022-35519-4, doi:10.1038/s41467-022-35519-4.
Matthew E. Ritchie, Belinda Phipson, Di Wu, Yifang Hu, Charity W. Law, Wei Shi, and Gordon K. Smyth. Limma powers differential expression analyses for term`rna`-sequencing and microarray studies. Nucleic acids research, 43(7):e47–e47, Apr 2015. gkv007[PII]. URL: https://doi.org/10.1093/nar/gkv007, doi:10.1093/nar/gkv007.
Mark D. Robinson, Davis J. McCarthy, and Gordon K. Smyth. Edger: a bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics (Oxford, England), 26(1):139–140, Jan 2010. btp616[PII]. URL: https://doi.org/10.1093/bioinformatics/btp616, doi:10.1093/bioinformatics/btp616.
Charlotte Soneson and Mark D. Robinson. Bias, robustness and scalability in single-cell differential expression analysis. Nature Methods, 15(4):255–261, Apr 2018. URL: https://doi.org/10.1038/nmeth.4612, doi:10.1038/nmeth.4612.
Jordan W. Squair, Matthieu Gautier, Claudia Kathe, Mark A. Anderson, Nicholas D. James, Thomas H. Hutson, Rémi Hudelle, Taha Qaiser, Kaya J. E. Matson, Quentin Barraud, Ariel J. Levine, Gioele La Manno, Michael A. Skinnider, and Grégoire Courtine. Confronting false discoveries in single-cell differential expression. Nature Communications, 12(1):5692, Sep 2021. URL: https://doi.org/10.1038/s41467-021-25960-2, doi:10.1038/s41467-021-25960-2.
Andrew L Thurman, Jason A Ratcliff, Michael S Chimenti, and Alejandro A Pezzulo. Differential gene expression analysis for multi-subject single cell term`RNA` sequencing studies with aggregateBioVar. Bioinformatics, 37(19):3243–3251, May 2021.
Catalina A. Vallejos, Davide Risso, Antonio Scialdone, Sandrine Dudoit, and John C. Marioni. Normalizing single-cell term`rna` sequencing data: challenges and opportunities. Nature Methods, 14(6):565–571, Jun 2017. URL: https://doi.org/10.1038/nmeth.4292, doi:10.1038/nmeth.4292.
Tianyu Wang, Boyang Li, Craig E. Nelson, and Sheida Nabavi. Comparative analysis of differential gene expression analysis tools for single-cell term`rna` sequencing data. BMC Bioinformatics, 20(1):40, Jan 2019. URL: https://doi.org/10.1186/s12859-019-2599-6, doi:10.1186/s12859-019-2599-6.
Kip D. Zimmerman, Mark A. Espeland, and Carl D. Langefeld. A practical solution to pseudoreplication bias in single-cell studies. Nature Communications, 12(1):738, Feb 2021. URL: https://doi.org/10.1038/s41467-021-21038-1, doi:10.1038/s41467-021-21038-1.
17.9. Contributors#
We gratefully acknowledge the contributions of:
